Advertisements
Advertisements
प्रश्न
Figure shows the variation in the internal energy U with the volume V of 2.0 mol of an ideal gas in a cyclic process abcda. The temperatures of the gas at b and c are 500 K and 300 K respectively. Calculate the heat absorbed by the gas during the process.

उत्तर
Given: Number of moles of the gas, n = 2 moles
The system's volume is constant for lines bc and da.
Therefore,
∆V = 0
Thus, work done for paths da and bc is zero.
⇒ Wda = Wbc = 0
Since the process is cyclic, ∆U is equal to zero.
Using the first law, we get
∆W = ∆Q
∆W = ∆WAB + ∆WCD
Since the temperature is kept constant during lines ab and cd, these are isothermal expansions.
Work done during an isothermal process is given by
W = nRT
\[\ln\frac{V_f}{V_i}\]
If Vf and Vi are the initial and final volumes during the isothermal process, then
\[W = n {RT}_1 ln\left( \frac{2 V_0}{V_0} \right) + n {RT}_2 ln\left( \frac{V_0}{2 V_0} \right)\]
W = nR × 2.303 × log 2 × (500 − 300)
W = 2 × 8.314 × 2.303 × 0.301 × 200
W = 2305.31 J
APPEARS IN
संबंधित प्रश्न
A solar cooker and a pressure cooker both are used to cook food. Treating them as thermodynamic systems, discuss the similarities and differences between them.
Answer the following in one or two sentences.
State the first law of thermodynamics.
A mixture of hydrogen and oxygen is enclosed in a rigid insulating cylinder. It is ignited by a spark. The temperature and pressure both increase considerably. Assume that the energy supplied by the spark is negligible, what conclusions may be drawn by application of the first law of thermodynamics?
ΔU is equal to ____________ work.
The process, in which no heat enters or leaves the system, is termed as ____________.
The isothermal bulk modulus of a perfect gas at pressure P is numerically equal to ____________.
Change in internal energy, when 4 KJ of work is done on the system and 1 KJ heat is given out by the system, is:
An ideal gas undergoes cyclic process ABCDA as shown in given P-V diagram (figure). The amount of work done by the gas is ______.

Consider a cycle followed by an engine (Figure)
1 to 2 is isothermal
2 to 3 is adiabatic
3 to 1 is adiabatic
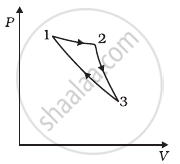
Such a process does not exist because ______.
- heat is completely converted to mechanical energy in such a process, which is not possible.
- mechanical energy is completely converted to heat in this process, which is not possible.
- curves representing two adiabatic processes don’t intersect.
- curves representing an adiabatic process and an isothermal process don’t intersect.
Consider one mole of perfect gas in a cylinder of unit cross section with a piston attached (figure). A spring (spring constant k) is attached (unstretched length L) to the piston and to the bottom of the cylinder. Initially the spring is unstretched and the gas is in equilibrium. A certain amount of heat Q is supplied to the gas causing an increase of volume from V0 to V1.

- What is the initial pressure of the system?
- What is the final pressure of the system?
- Using the first law of thermodynamics, write down a relation between Q, Pa, V, Vo and k.
Write the mathematical equation for the first law of thermodynamics for:
Isothermal process
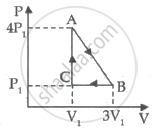
An ideal gas is taken through series of changes ABCA. The amount of work involved in the cycle is ______.
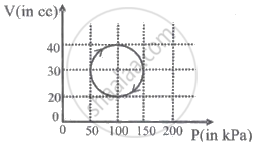
A system is taken through a cyclic process represented by a circle as shown. The heat absorbed by the system is ______.
ΔU = 0 is true for ______.
104 J of work is done on a certain volume of a gas. If the gas releases 125 kJ of heat, calculate the change in internal energy of the gas.
In an adiabatic expansion of 2 moles of a gas, the initial pressure was 1.013 × 105 Pa, the initial volume was 22.4 L, the final pressure was 3.191 × 104 Pa and the final volume was 44.8 L. Find the work done by the gas on its surroundings. Taken `γ = 5/3`.
Consider the cyclic process ABCA on a sample of 2.0 mol of an ideal gas as shown in following figure. The temperature of the gas at A and B are 300 K and 500 K respectively. A total of 1200 J heat is withdrawn from the sample in this process. Find the work done by the gas in part BC. (R = 8.3 J/mol K)
Show that the heat absorbed at constant pressure is equal to the change in enthalpy of the system.
