Advertisements
Advertisements
प्रश्न
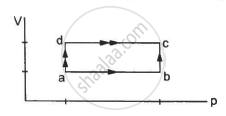
An ideal gas undergoes cyclic process ABCDA as shown in given P-V diagram (figure). The amount of work done by the gas is ______.

विकल्प
6P0V0
–2P0V0
+ 2P0V0
+ 4P0V0
उत्तर
An ideal gas undergoes cyclic process ABCDA as shown in given P-V diagram (figure). The amount of work done by the gas is `underline(- 2P_0V_0)`.
Explanation:
Consider the P-V diagram given in the question.
Work done in the process ABCD = Area of rectangle ABCDA
= (AB) × BC
= (3V0 – V0) × (2P0 – P0)
= 2V0 × P0
= 2p0V0
As the process is going anticlockwise, hence there is a net compression in the gas. So, work done by the gas = –2P0V0.
APPEARS IN
संबंधित प्रश्न
The internal energy of an ideal gas decreases by the same amount as the work done by the system.
(a) The process must be adiabatic.
b) The process must be isothermal.
(c) The process must be isobaric.
(d) The temperature must decrease.
When a system is taken through the process abc shown in figure, 80 J of heat is absorbed by the system and 30 J of work is done by it. If the system does 10 J of work during the process adc, how much heat flows into it during the process?
Define an isolated system.
"The mass and energy both are conserved in an isolated system", is the statement of ______.
Three copper blocks of masses M1, M2 and M3 kg respectively are brought into thermal contact till they reach equilibrium. Before contact, they were at T1, T2, T3 (T1 > T2 > T3). Assuming there is no heat loss to the surroundings, the equilibrium temprature T is (s is specific heat of copper)
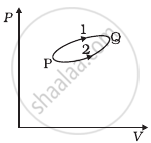
A system goes from P to Q by two different paths in the P-V diagram as shown in figure. Heat given to the system in path 1 is 1000 J. The work done by the system along path 1 is more than path 2 by 100 J. What is the heat exchanged by the system in path 2?
The first law of thermodynamics for isothermal process is ______.
The amount of work done in increasing the voltage across the plates of capacitor from 5 V to 10 V is W. The work done in increasing it from 10 V to 15 V will be ______.
What is true for an adiabatic process?
Choose the correct relation with reason.
