Advertisements
Advertisements
प्रश्न
An ideal gas undergoes cyclic process ABCDA as shown in given P-V diagram (figure). The amount of work done by the gas is ______.

पर्याय
6P0V0
–2P0V0
+ 2P0V0
+ 4P0V0
उत्तर
An ideal gas undergoes cyclic process ABCDA as shown in given P-V diagram (figure). The amount of work done by the gas is `underline(- 2P_0V_0)`.
Explanation:
Consider the P-V diagram given in the question.
Work done in the process ABCD = Area of rectangle ABCDA
= (AB) × BC
= (3V0 – V0) × (2P0 – P0)
= 2V0 × P0
= 2p0V0
As the process is going anticlockwise, hence there is a net compression in the gas. So, work done by the gas = –2P0V0.
APPEARS IN
संबंधित प्रश्न
Write the mathematical expression of the First Law of Thermodynamics for Isothermal Process
Calculate the change in internal energy of a gas kept in a rigid container when 100 J of heat is supplied to it.
When a system is taken through the process abc shown in figure, 80 J of heat is absorbed by the system and 30 J of work is done by it. If the system does 10 J of work during the process adc, how much heat flows into it during the process?
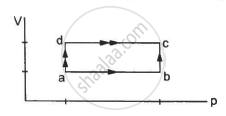
Calculate the heat absorbed by a system in going through the cyclic process shown in figure.
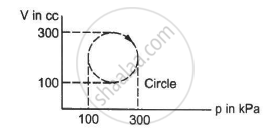
The internal energy of a gas is given by U = 1.5 pV. It expands from 100 cm3 to 200 cm3against a constant pressure of 1.0 × 105 Pa. Calculate the heat absorbed by the gas in the process.
The compressibility of water is 5 × 10-10 m2/N. Pressure of 15 × 106 Pa is applied on 100 ml volume of water. The change in the volume of water is ______.
Consider a P-V diagram in which the path followed by one mole of perfect gas in a cylindrical container is shown in figure.
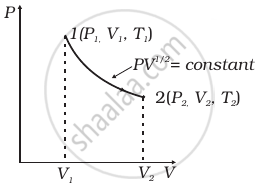
- Find the work done when the gas is taken from state 1 to state 2.
- What is the ratio of temperature T1/T2, if V2 = 2V1?
- Given the internal energy for one mole of gas at temperature T is (3/2) RT, find the heat supplied to the gas when it is taken from state 1 to 2, with V2 = 2V1.
Write the mathematical equation for the first law of thermodynamics for:
Isothermal process
An insulated container of gas has two chambers separated by an insulating partition. One of the chambers has volume V1 and contains ideal gas at pressure P1 and temperature T1. The other chamber has volume V2 and contains ideal gas at pressure P2 and temperature T2. If the partition is removed without doing any work on the gas, the final equilibrium temperature of the gas in the container will be ______.
Explain the formulation of first law of thrmodynamics.
