Advertisements
Advertisements
प्रश्न
Consider two containers A and B containing identical gases at the same pressure, volume and temperature. The gas in container A is compressed to half of its original volume isothermally while the gas in container B is compressed to half of its original value adiabatically. The ratio of final pressure of gas in B to that of gas in A is ______.
पर्याय
`2^(γ - 1)`
`(1/2)^(γ - 1)`
`(1/(1 - γ))^2`
`(1/(γ - 1))^2`
उत्तर
Consider two containers A and B containing identical gases at the same pressure, volume and temperature. The gas in container A is compressed to half of its original volume isothermally while the gas in container B is compressed to half of its original value adiabatically. The ratio of final pressure of gas in B to that of gas in A is `underline(2^(γ - 1))`.
Explanation:
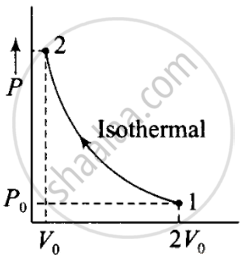
According to the P-V diagram is shown for container A (which is going through an isothermal process) and for container B (which is going through an adiabatic process).
V →container A

V →container A
Both processes involve compression of the gas.
(i) Isothermal compression (gas A) (during 1→ 2)
P1V1 = P2V2
⇒ P0(2V0)γ = P2(V0)γ
⇒ P0(2V0) = P2(V0)
(ii) Adiabatic compression, (gas B) (during 1→ 2)
P1V1γ = P2V2γ
⇒ P0(2V0)γ = P2(V0)γ
⇒ P2 = `((2V_0)/V_0) P_0 - (2)^γ P_0`
Hence `((P_2)_B)/((P_2)_A)` = ratio of final pressure = `((2)^γ P_0)/(2P_0) = 2^(γ - 1)` where, γ is ratio of specific heat capacities for the gas.
APPEARS IN
संबंधित प्रश्न
"The mass and energy both are conserved in an isolated system", is the statement of ______.
A sample of gas absorbs 4000 kJ of heat and surrounding does 2000 J of work on sample, what is the value of ΔU?
Is it possible to increase the temperature of a gas without adding heat to it? Explain.
Write the mathematical equation for the first law of thermodynamics for:
Adiabatic process
In the reported figure, heat energy absorbed by a system in going through a cyclic process is ______ πJ.
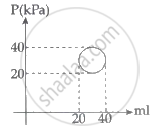
A system is taken through a cyclic process represented by a circle as shown. The heat absorbed by the system is ______.
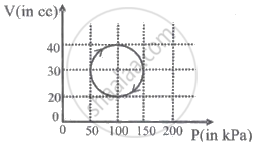
An ideal gas (γ = 1.5) is expanded adiabatically. How many times has the gas had to be expanded to reduce the root mean square velocity of molecules two times?
Show that the heat absorbed at constant pressure is equal to the change in enthalpy of the system.
Obtain an expression for the workdone by a gas in an isothermal process.
Define isochoric process
