Advertisements
Advertisements
Question
Consider two containers A and B containing identical gases at the same pressure, volume and temperature. The gas in container A is compressed to half of its original volume isothermally while the gas in container B is compressed to half of its original value adiabatically. The ratio of final pressure of gas in B to that of gas in A is ______.
Options
`2^(γ - 1)`
`(1/2)^(γ - 1)`
`(1/(1 - γ))^2`
`(1/(γ - 1))^2`
Solution
Consider two containers A and B containing identical gases at the same pressure, volume and temperature. The gas in container A is compressed to half of its original volume isothermally while the gas in container B is compressed to half of its original value adiabatically. The ratio of final pressure of gas in B to that of gas in A is `underline(2^(γ - 1))`.
Explanation:
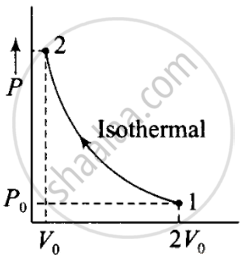
According to the P-V diagram is shown for container A (which is going through an isothermal process) and for container B (which is going through an adiabatic process).
V →container A

V →container A
Both processes involve compression of the gas.
(i) Isothermal compression (gas A) (during 1→ 2)
P1V1 = P2V2
⇒ P0(2V0)γ = P2(V0)γ
⇒ P0(2V0) = P2(V0)
(ii) Adiabatic compression, (gas B) (during 1→ 2)
P1V1γ = P2V2γ
⇒ P0(2V0)γ = P2(V0)γ
⇒ P2 = `((2V_0)/V_0) P_0 - (2)^γ P_0`
Hence `((P_2)_B)/((P_2)_A)` = ratio of final pressure = `((2)^γ P_0)/(2P_0) = 2^(γ - 1)` where, γ is ratio of specific heat capacities for the gas.
APPEARS IN
RELATED QUESTIONS
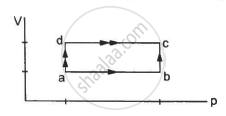
When a system is taken through the process abc shown in figure, 80 J of heat is absorbed by the system and 30 J of work is done by it. If the system does 10 J of work during the process adc, how much heat flows into it during the process?
Find the change in the internal energy of 2 kg of water as it is heated from 0°C to 4°C. The specific heat capacity of water is 4200 J kg−1 K−1 and its densities at 0°C and 4°C are 999.9 kg m−3 and 1000 kg m−3 respectively. Atmospheric pressure = 105 Pa.
A sample of gas absorbs 4000 kJ of heat and surrounding does 2000 J of work on sample. What is the value of ∆U?
When heat energy of 2000 joules is supplied to a gas at constant pressure 2.1 x 105 N/m2, there is an increase in its volume equal to 2.5 x 10-3 m3. The increase in internal energy of the gas in joules is ____________.
A gas performs 0.320 kJ work on surrounding and absorbs 120 J of heat from the surrounding. Hence, change in internal energy is ______.
The isothermal bulk modulus of a perfect gas at pressure P is numerically equal to ____________.
Derive an expression for the work done during an isothermal process.
For an isothermal and reversible expansion of 0.5 mol of an ideal gas Wmax is - 3.918 kJ. The value of ΔU is ______.
A monoatomic gas at 27°C is adiabatically compressed to 80% of its initial volume. Find the final temperature of the gas.
Define the isothermal process.
