Advertisements
Advertisements
प्रश्न
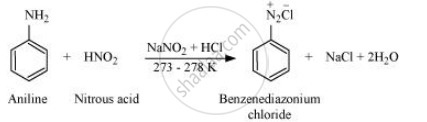
Write the reaction of aromatic primary amine with nitrous acid.
उत्तर
Aromatic amine react with nitrous acid (prepared in situ from NaNO2 and a mineral acid such as HCl) at 273 − 278 K to form stable aromatic diazonium salts, i.e., NaCl and H2O.
APPEARS IN
संबंधित प्रश्न
Write a short note on the following:
Hoffmann’s bromamide reaction
Give the structure of A, B and C in the following reaction:
\[\ce{C6H5N2Cl ->[CuCN] A ->[H2O/H+] B ->[NH3][\Delta] C}\]
Explain the mechanism of action of hydroiodic acid on 3-methylbutan-2-ol.
Arrange the following in the increasing order of their pKb values:
C6H5NH2, C2H5NH2, C6H5NHCH3
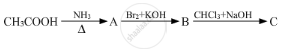
Give the structures of A, B and C in the following reactions :
Identify the product 'A' in the following reaction.
\[\ce{Aniline ->[(CH3CO)2O][Pyridine] A}\]

Identify the major product (B).
Identify product B in the following reaction.
\[\ce{Aniline ->[NaNO2][HCl] A ->[KI] B}\]
Which of the following reactions does NOT yield an amine?
Which of the following compounds is the weakest Brönsted base?
Which of the following amines can be prepared by Gabriel synthesis.
(i) Isobutyl amine
(ii) 2-Phenylethylamine
(iii) N-methylbenzylamine
(iv) Aniline
Under which of the following reaction conditions, aniline gives p-nitro derivative as the major product?
(i) Acetyl chloride/pyridine followed by reaction with conc.\[\ce{H2SO4 }\] + conc. \[\ce{HNO3}\].
(ii) Acetic anyhdride/pyridine followed by conc.\[\ce{H2SO4}\] + conc.\[\ce{HNO3}\].
(iii) Dil. HCl followed by reaction with conc.\[\ce{H2SO4}\] + conc.\[\ce{HNO3}\].
(iv) Reaction with conc.\[\ce{HNO3}\] + conc.\[\ce{H2 SO4}\].
Write following conversions:
acetanilide `->` p-nitroaniline
Methyl amine on reaction with chloroform in the presence of NaOH gives ______.
The amine 'A' when treated with nitrous acid gives yellow oily substance. The amine A is ______.
What is the IUPAC name of \[\ce{(CH3)2 - N - CH3}\]?
Which of the following would not be a good choice for reducing nitrobenzene to aniline?
Assertion: Amimonolysis of alkyl halides involves the reaction between alkyl halides and alcoholic ammonia.
Reason: Ammonolysis of alkyl halides produces secondary amines only.
