Advertisements
Advertisements
प्रश्न
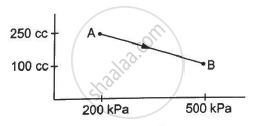
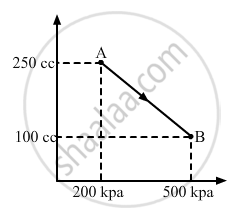
A gas is taken along the path AB as shown in figure. If 70 cal of heat is extracted from the gas in the process, calculate the change in the internal energy of the system.
उत्तर
Given:- 70 cal of heat is extracted from the system.
Here,
∆Q = -70 cal = -(70 × 4.2) J = -294 J
From the first law of thermodynamics, we get
∆W = P ∆ V
If P is the average pressure between points A and B and ∆V is the change in volume of the system while going from point A to B, then
∆W = `-1/2`× (200 + 500) × 103 × (150 × 10−6)
∆W = `-1/2`× 700 × 150 × 10−3
∆W = - 525 × 10−1 = - 52.5 J
Here, negative sign is taken because the final volume is less than the initial volume.
∆U = ?
∆Q = ∆U + ∆W
∆Q = - 294 J
Here, negative sign indicates that heat is extracted out from the system.
⇒ − 294 = ∆U - 52.5
⇒ ∆U = − 294 + 52.5 = - 241.5 J
APPEARS IN
संबंधित प्रश्न
A steam engine delivers 5.4×108 J of work per minute and services 3.6 × 109 J of heat per minute from its boiler. What is the efficiency of the engine? How much heat is wasted per minute?
A cylinder containing a gas is lifted from the first floor to the second floor. What is the amount of work done on the gas? What is the amount of work done by the gas? Is the internal energy of the gas increased? Is the temperature of the gas increased?
A force F is applied on a block of mass M. The block is displaced through a distance d in the direction of the force. What is the work done by the force on the block? Does the internal energy change because of this work?
When we rub our hands they become warm. Have we supplied heat to the hands?
Consider the following two statements.
(A) If heat is added to a system, its temperature must increase.
(B) If positive work is done by a system in a thermodynamic process, its volume must increase.
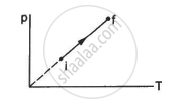
An ideal gas goes from the state i to the state f as shown in figure. The work done by the gas during the process ______________ .
The pressure p and volume V of an ideal gas both increase in a process.
(a) Such a process is not possible.
(b) The work done by the system is positive.
(c) The temperature of the system must increase.
(d) Heat supplied to the gas is equal to the change in internal energy.
Which of the following system freely allows the exchange of energy and matter with its environment?
What is the internal energy of the system, when the amount of heat Q is added to the system and the system does not do any work during the process?
When does a system lose energy to its surroundings and its internal energy decreases?
Explain given cases related to energy transfer between the system and surrounding –
- energy transferred (Q) > 0
- energy transferred (Q) < 0
- energy transferred (Q) = 0
A cylinder containing one gram molecule of the gas was compressed adiabatically until its temperature rose from 27°C to 97°C. Calculate the work done and heat produced in the gas (𝛾 = 1.5).
8 m3 of a gas is heated at the pressure 105 N/m2 until its volume increases by 10%. Then, the external work done by the gas is ____________.
Which of the following represents isothermal process?
Two cylinders A and B of equal capacity are connected to each other via a stopcock. A contains a gas at standard temperature and pressure. B is completely evacuated. The entire system is thermally insulated. The stopcock is suddenly opened. Answer the following:
What is the change in internal energy of the gas?
In insulated systems, the amount of external work done by the gas is proportional to:
Figure shows the P-V diagram of an ideal gas undergoing a change of state from A to B. Four different parts I, II, III and IV as shown in the figure may lead to the same change of state.

- Change in internal energy is same in IV and III cases, but not in I and II.
- Change in internal energy is same in all the four cases.
- Work done is maximum in case I
- Work done is minimum in case II.
The internal energy of one mole of argon is ______.
The molar specific heat of He at constant volume is 12.47 J/mol.K. Two moles of He are heated at constant pressure. So the rise in temperature is 10 K. Find the increase in internal energy of the gas.
