Advertisements
Advertisements
प्रश्न
An ideal gas is trapped between a mercury column and the closed-end of a narrow vertical tube of uniform base containing the column. The upper end of the tube is open to the atmosphere. The atmospheric pressure equals 76 cm of mercury. The lengths of the mercury column and the trapped air column are 20 cm and 43 cm respectively. What will be the length of the air column when the tube is tilted slowly in a vertical plane through an angle of 60°? Assume the temperature to remain constant.
उत्तर
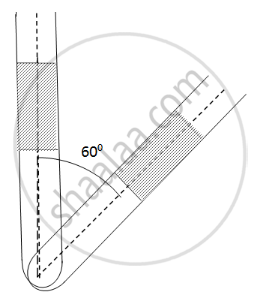
Here ,
Initial pressure = Atmospheric pressure + pressure due to mercury
⇒ P1 = P0 + PHg
Let the CSA of the tube be A.
P1 = 0.76 + 0.2 = 0.96 m Hg
T1 = T2 = T
V1 = 0.43 A
If the tube is slanted , then the atmospheric pressure P0 remains the same . only the PHg changes
`P_2 = P_0 + P_Hg cos60^circ` = 0.76 + 0.2 × 0.5 = 0.86
`P_1V_1 = P_2V_2`
⇒ `V_2 = (P_1V_1)/P_2 = (0.96×0.43A)/0.86`
Let the length of the air column be l.
⇒ Al = `(P_1V_1)/P_2 = (0.96×0.43A)/0.86`
⇒ l = 0.48 m
⇒ l = 48 cm
APPEARS IN
संबंधित प्रश्न
Consider a gas of neutrons. Do you expect it to behave much better as an ideal gas as compared to hydrogen gas at the same pressure and temperature?
If the molecules were not allowed to collide among themselves, would you expect more evaporation or less evaporation?
The pressure of an ideal gas is written as \[P = \frac{2E}{3V}\] . Here E refers to
The mean square speed of the molecules of a gas at absolute temperature T is proportional to
The temperature and pressure at Simla are 15.0°C and 72.0 cm of mercury and at Kalka these are 35.0°C and 76.0 cm of mercury. Find the ratio of air density at Kalka to the air density at Simla.
Use R=8.314J K-1 mol-1
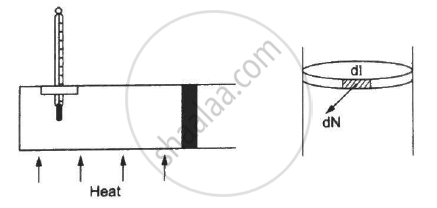
Figure shows a cylindrical tube of radius 5 cm and length 20 cm. It is closed by a tight-fitting cork. The friction coefficient between the cork and the tube is 0.20. The tube contains an ideal gas at a pressure of 1 atm and a temperature of 300 K. The tube is slowly heated and it is found that the cork pops out when the temperature reaches 600 K. Let dN denote the magnitude of the normal contact force exerted by a small length dlof the cork along the periphery (see the figure). Assuming that the temperature of the gas is uniform at any instant, calculate `(dN)/(dt)`.
The condition of air in a closed room is described as follows. Temperature = 25°C, relative humidity = 60%, pressure = 104 kPa. If all the water vapour is removed from the room without changing the temperature, what will be the new pressure? The saturation vapour pressure at 25°C − 3.2 kPa.
Answer in brief:
Show that rms velocity of an oxygen molecule is `sqrt2` times that of a sulfur dioxide molecule at S.T.P.
In an ideal gas, the molecules possess
Compare the rate of radiation of metal bodies at 727 °C and 227 °C.
What is the microscopic origin of temperature?
Explain in detail the kinetic interpretation of temperature.
A ring of mass m and radius r rotates about an axis passing through its centre and perpendicular to its plane with angular velocity `omega`. Its kinetic energy is ______.
A molecule consists of two atoms each of mass 'm' and separated by a distance 'd'. At room temperature the average rotational kinetic energy is 'E', then its angular frequency is ______.
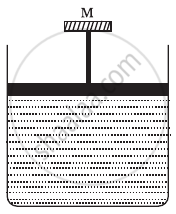
A cylinder containing an ideal gas is in vertical position and has a piston of mass M that is able to move up or down without friction (Figure). If the temperature is increased ______.
23Ne decays to 23Na by negative beta emission. Mass of 23Ne is 22.994465 amu mass of 23Na is 22.989768 amu. The maximum kinetic energy of emitted electrons neglecting the kinetic energy of recoiling product nucleus is ______ MeV.
According to the kinetic theory of gases, at a given temperature, molecules of all gases have the same ______.
If a = 0. 72 and t = 0.04, then the value of r is ______.
Which of the following materials is diathermanous?
