Advertisements
Advertisements
प्रश्न
Define the following terms:
Pseudo first-order reaction
उत्तर
Pseudo first-order reaction
Pseudo first-order reaction: The reaction which is bimolecular but whose order is one is called pseudo first-order reaction. This happens when one of the reactants is present in a large amount. For example, in acidic hydrolysis of ester (ethyl acetate), water is present in a large quantity.
CH3COOC2H5 + H2O →→ CH3COOH + C2H5OH
Similarly, inversion of cane sugar or hydrolysis of cane sugar is an example of pseudo first-order reaction.
APPEARS IN
संबंधित प्रश्न
Write molecularity of the following reaction:
2NO(g)+O2(g)→2NO2(g)
For a chemical reaction R → P, the variation in the concentration (R) vs. time (t) plot is given as:
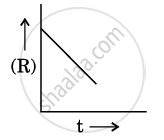
(i) Predict the order of the reaction.
(ii) What is the slope of the curve ?
(iii) Write the unit of rate constant for this reaction.
Which of the following statements is not correct about order of a reaction.
Why can we not determine the order of a reaction by taking into consideration the balanced chemical equation?
Use Molecular Orbital theory to determine the bond order in each of species, [He2j+ and [He2]2+?
A catalyst in a reaction changes which of the following?
For a reaction A + B → products, the rate law is given by: r = `K[A]^(1/2)`. What is the order of reaction?
A drop of solution (volume 0.05 ml) contains 3.0 × 10-6 mole of H+. If the rate constant of disappearance of H+ is 1.0 × 107 mole l-1s-1. It would take for H+ in drop to disappear in ______ × 10-9s.
Which of the following statement is true?
A reaction is second order with respect to a reactant. How is the rate of reaction affected if the concentration of the reactant is reduced to half?
