Advertisements
Advertisements
Question
Define the following terms:
Pseudo first-order reaction
Solution
Pseudo first-order reaction
Pseudo first-order reaction: The reaction which is bimolecular but whose order is one is called pseudo first-order reaction. This happens when one of the reactants is present in a large amount. For example, in acidic hydrolysis of ester (ethyl acetate), water is present in a large quantity.
CH3COOC2H5 + H2O →→ CH3COOH + C2H5OH
Similarly, inversion of cane sugar or hydrolysis of cane sugar is an example of pseudo first-order reaction.
APPEARS IN
RELATED QUESTIONS
For a reaction : 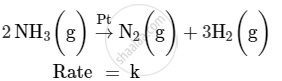
(i) Write the order and molecularity of this reaction.
(ii) Write the unit of k.
For a chemical reaction R → P, the variation in the concentration (R) vs. time (t) plot is given as:
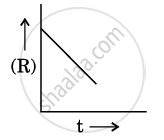
(i) Predict the order of the reaction.
(ii) What is the slope of the curve ?
(iii) Write the unit of rate constant for this reaction.
From the rate expression for the following reaction, determine the order of reaction and the dimension of the rate constant.
\[\ce{C2H5Cl_{(g)} -> C2H4_{(g)} + HCl_{(g)}}\] Rate = k [C2H5Cl]
Write resonating structures of ozone.
Which of the following statements is not correct about order of a reaction.
For a complex reaction:
(i) order of overall reaction is same as molecularity of the slowest step.
(ii) order of overall reaction is less than the molecularity of the slowest step.
(iii) order of overall reaction is greater than molecularity of the slowest step.
(iv) molecularity of the slowest step is never zero or non interger.
Why molecularity is applicable only for elementary reactions and order is applicable for elementary as well as complex reactions?
Assertion: Order and molecularity are same.
Reason: Order is determined experimentally and molecularity is the sum of the stoichiometric coefficient of rate determining elementary step.
For a reaction A + B → products, the rate law is given by: r = `K[A]^(1/2)`. What is the order of reaction?
If the 0.05 molar solution of m+ is replaced by a 0.0025 molar m+ solution, then the magnitude of the cell potential would be
