Advertisements
Advertisements
प्रश्न
Draw a p-V diagram of the reversible process.
उत्तर
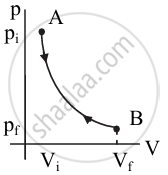
p-V diagram of reversible process
संबंधित प्रश्न
A thermodynamic system is taken from an original state to an intermediate state by the linear process shown in Figure

Its volume is then reduced to the original value from E to F by an isobaric process. Calculate the total work done by the gas from D to E to F
Give an example of some familiar process in which heat is added to an object, without changing its temperature.
For work done to be reversible, the process should be ______
Draw a p-V diagram showing positive work at constant pressure.
Differentiate between the reversible and irreversible processes.
3 mole of a gas at temperature 400 K expands isothermally from an initial volume of 4 litres to a final volume of 8 litres. Find the work done by the gas. (R = 8.31 J mol-1 K-1)
Explain the thermodynamics of the isobaric process.
Explain thermodynamics of the adiabatic process.
When you exercise in the morning, by considering your body as a thermodynamic system, which of the following is true?
Apply first law for an adiabatic process.
Draw the PV diagram for the isochoric process.
Can the given heat energy be completely converted to work in a cyclic process? If not, when can the heat can completely converted to work?
Explain in detail the isochoric process.
In a petrol engine, (internal combustion engine) air at atmospheric pressure and temperature of 20°C is compressed in the cylinder by the piston to `1/8` of its original volume. Calculate the temperature of the compressed air. (For air γ = 1.4)
A monoatomic gas of pressure p having volume V expands isothermally to a volume 2V and then adiabatically to a volume 16V. The final pressure of the gas is ____________.
`("ratio of specific heats" = 5/3)`
Assertion: Equal volumes of monatomic and polyatomic gases are adiabatically compressed separately to equal compression ratio `("P"_2/"P"_1)`. Then monatomic gas will have greater final volume.
Reason: Among ideal gases, molecules of a monatomic gas have the smallest number of degrees of freedom.
Ideal gas for which 'ϒ' = 1.5 is suddenly compressed to `1/4`th of its initial volume. The ratio of 4 the final pressure to the initial pressure is ______.
`(ϒ = "C"_"p"/"C"_"v")`
Consider P-V diagram for an ideal gas shown in figure.
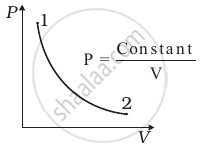
Out of the following diagrams (figure), which represents the T-P diagram?
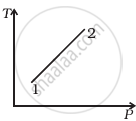 (i) |
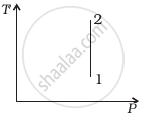 (ii) |
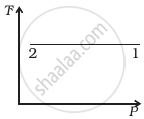 (iii) |
 (iv) |
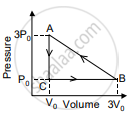
In the figure shown here, the work done in the process ACBA is ______.
In a certain thermodynamical process, the pressure of a gas depends on its volume as kV3. The work done when the temperature changes from 100°C to 300°C will be ______ nR, where n denotes number of moles of a gas.
Explain the thermodynamic process.
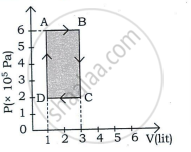
An ideal gas is taken through a cyclic process ABCDA as shown in figure. The net work done by the gas during the cycle is ______.
Explain how can a gas be expanded at constant temperature.
