Advertisements
Advertisements
प्रश्न
Draw the geometrical isomers of the following complexes [Co(NH3)4Cl2]+
उत्तर
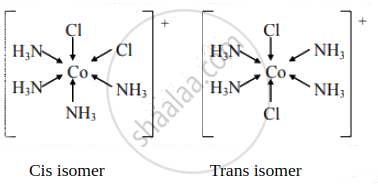
संबंधित प्रश्न
List various types of isomerism possible for coordination compounds, giving an example of each.
Out of 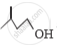 and
and 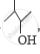 , which one is optically active and why ?
, which one is optically active and why ?
Choose the most correct option.
Which of the following complexes exist as cis and trans isomers?
1. [Cr(NH3)2Cl4]-
2. [Co(NH3)5Br]2⊕
3. [PtCl2Br2]2- (square planar)
4. [FeCl2(NCS)2]2- (tetrahedral)
Answer the following in one or two sentences.
Consider the complexes \[\ce{[Cu(NH3)4][PtCl4] and [Pt(NH3)4] [CuCl4]}\]. What type of isomerism these two complexes exhibit?
Answer in brief.
What are ionization isomers ? Give an example.
Draw isomers of the following.
\[\ce{Pt(NH3)2ClNO2}\]
Write the type of isomerism exhibited by [Co(NH3)5(NO2)]2+ and [Co(NH3)5ONO]2+ pair of complex ion.
Draw optical isomers of [Co(en)3]3+.
Which one of the following will give a pair of enantiomorphs?
Fac-mer isomerism is shown by
The complex ions [Co(H2O)5(ONO)]2+ and [Co(H2O)5NO2]2+ are ____________.
The formula of two complexes X and Y of chromium are given below:
\[\ce{\underset{(X)}{[Cr(H2O)6]Cl3}}\] and \[\ce{\underset{(Y)}{[Cr(H2O)5Cl]Cl2.H2O}}\]
X and Y are examples of ____________ isomers.
Which of the following has zero dipole moment?
What is the number of moles of silver chloride precipitated when excess of aqueous silver nitrate is treated with [Co(NH3)4Cl2]Cl?
Complex [COCl2(en)2]+ can
Which of the following has an optical isomer?
Which of the following shows maximum number of isomers?
\[\ce{CH3CH2COO- Na+ ->[NaOH, + ?][Heat] CH3CH3 + Na2CO3}\]
Consider the above reaction and identify the missing reagent/chemical.
Which among the following solid is a non-polar solid?
Give cis isomer of [Co(NH3)4Cl2]⊕.
Indicate the type of isomerism exhibited by the following complex and draw the structures for this isomer:
\[\ce{[Pt(NH3)(H2O)Cl2]}\]
Explain the geometrical isomerism of the octahedral complex of the type [M(AA)2B2]n± with a suitable example.
Which one of the following complex ions has geometrical isomers?
Three organic compounds A, B and C are non cyclic functional isomers of carbonyl compounds with molecular formula C4H8O. Isomers A and C give positive Tollen’s test while compound B does not give positive Tollen’s test but gives positive iodoform test. Compounds A and B on reduction with Zn amalgam and conc. HCl give the same product.
- Write the structures of the compounds A, B and C.
- Out of the compounds A, B and C, which one will be the least reactive towards addition of HCN.
