Advertisements
Advertisements
प्रश्न
Find the kinetic energy of the emitted electron, if in photoelectric effect energy of incident Photon is 4 eV and work function is 2.4 eV.
उत्तर
K.E.max = hν − φ0
= 4 − 2.4
= 1.6 eV.
APPEARS IN
संबंधित प्रश्न
Choose the correct option.
Polychromatic (containing many different frequencies) radiation is used in an experiment on the photoelectric effect. The stopping potential ______.
Explain the inverse linear dependence of stopping potential on the incident wavelength in a photoelectric effect experiment.
The threshold wavelength of tungsten is 2.76 x 10-5 cm.
(a) Explain why no photoelectrons are emitted when the wavelength is more than 2.76 x 10-5 cm.
(b) What will be the maximum kinetic energy of electrons ejected in each of the following cases
(i) if ultraviolet radiation of wavelength λ = 1.80 × 10-5 cm and
(ii) radiation of frequency 4 x 1015 Hz is made incident on the tungsten surface?
As the intensity of incident light increases ______
If the total energy of radiation of frequency 1014 Hz is 6.63 J, Calculate the number of photons in the radiation.
State Einstein photoelectric equation. Explain 2 characteristics of the photoelectric effect on the basis of Einstein’s photoelectric equation.
With the help of a circuit diagram describing an experiment to study the photoelectric effect.
What is the photoelectric effect? Define stopping potential and photoelectric work function.
The energy of a photon is 2 eV. Find its frequency and wavelength.
With the help of a circuit diagram describe the experiment to study the characteristics of the photoelectric effect. Hence discuss any 2 characteristics of the photoelectric effect.
If the maximum kinetic energy of emitted electrons in photoelectric effect is 3.2 × 10-19 J and the work-function for metal is 6.63 × 10-19 J, then stopping potential and threshold wavelength respectively are
[Planck's constant, h = 6.63 × 1034 J-s]
[Velocity of light, c = 3 × 108 `"m"/"s"`]
[Charge on electron= 1.6 × 10-19 C]
The maximum velocity of the photoelectron emitted by the metal surface is 'v '. Charge and mass of the photoelectron is denoted by 'e' and 'm' respectively. The stopping potential in volt is ______.
A metal surface is illuminated by photons of energy 5 eV and 2.5 eV respectively. The ratio of their wavelengths is ____________.
When a surface 1 cm thick is illuminated by light of wavelength 'λ', the stopping potential is 'V0'. When the same surface is illuminated by light of wavelength '3λ', the stopping potential is `"V"_0/6`. The threshold wavelength for the metallic surface is ______.
The work function of a photosensitive material is 4.0 eV. The longest wavelength of light that can cause photon emission from the substance is (approximately) ____________.
Two incident radiations having energies two times and ten times of the work function of a metal surface, produce photoelectric effect. The ratio of maximum velocities of emitted photo electrons respectively is ____________.
When light of wavelength '`lambda`' is incident on photosensitive surface, photons of power 'P' are emitted. The number of photons (n) emitted in 't' second is (h = Planck's constant, c = velocity of light in vacuum) ____________.
A metal surface having work function 'w0' emits photoelectrons when photons of energy 'E' are incident on it. The electron enters the uniform magnetic field (B) in perpendicular direction and moves in circular path of radius 'r'. Then 'r' is equal to (m and e be the mass and charge of electron respectively) ____________.
Is it always necessary to use red light to get a photoelectric effect?
The wavelength of light incident on a metal surface is reduced from 300 nm to 200 nm (both are less than threshold wavelength). What is the change in the stopping potential for photoelectrons emitted from the surface will be ______ V. (Take h = 6.6 × 10-34 J-s)
The maximum kinetic energy of the photoelectrons ejected will be ______ eV when the light of wavelength 350 nm is incident on a cesium surface. The work function of cesium = 1.9 eV.
A charged dust particle of radius 5 × 10-7 m is located in a horizontal electric field having an intensity of 6.28 × 105 V/m. The surrounding medium is air with a coefficient of viscosity η = 1.6 × 10-5 N-s/m2. If the particle moves with a uniform horizontal speed of 0.02 m/s, the number of electrons on it is ______.
In a photocell, frequency of incident radiation is increased by keeping other factors constant (v > v0), the stopping potential ______.
If the electron in hydrogen atom jumps from second Bohr orbit to ground state and difference between energies of the two states is radiated in the form of photons. If the work function of the material is 4.2 eV, then stopping potential is ______.
[Energy of electron in nth orbit = `-13.6/"n"^2` eV ]
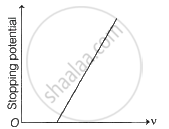
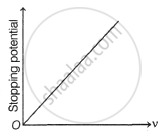
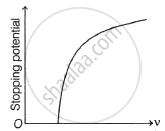
The following graphs show the variation of stopping potential corresponding to the frequency of incident radiation (ν) for a given metal. The correct variation is shown in graph [ν0 = threshold frequency].
|
(A) |
(B) |
|
(C) |
(D) |
Photoelectric emission is observed from a metallic surface for frequencies ν1 and ν2 of the incident light rays (ν1 > ν2). If the ratio of the maximum value of the kinetic energy of the photoelectrons emitted in the first case to that in the second case is 2 : K, then the threshold frequency of the metallic surface is ______.
By increasing the voltage in an electron diffraction tube, the radius of the diffraction rings will ______.