Advertisements
Advertisements
प्रश्न
If soap has high alkali content it irritates skin. How can the amount of excess alkali be determined? What can be the source of excess alkali?
उत्तर
Acid-base titration can be used to determine the excess amount of alkali in soap. The excess alkali left after hydrolysis of oils or fats can be the source of alkalinity in soap.
APPEARS IN
संबंधित प्रश्न
Explain cationic detergents.
Write the chemical equation for preparing sodium soap from Glyceryl palmitate . Structural formulae of the compounds are given below.
(C15H31COO)3C3H5 – Glyceryl palmitate
Explain the mechanism of cleansing action of soaps.
Write balanced chemical equations for the action of methyl bromide on silver propanoate
How soap is prepared?
Why is it safer to use soap from the environmental point of view?
What is the side product of soap industry? Give reactions showing soap formation.
What is the difference between bathing soap and washing soaps?
Match the detergents given in Column I with their uses given in Column II.
| Column I | Column II |
(i) 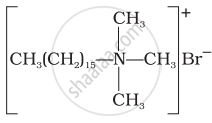 |
(a) Dishwashing powder |
(ii) 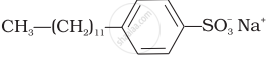 |
(b) Laundry soap |
| (iii) \[\ce{C17H33CO\overset{-}{O}\overset{+}{N}a + Na2CO3 + Rosin}\] | (c) Hair conditioners |
| (iv) \[\ce{CH3(CH2)16COO(CH2CH2O)nCH2CH2OH}\] | (d) Toothpaste |
Green chemistry in day-to-day life is in the use of ______.
