Advertisements
Advertisements
प्रश्न
One chemical test that would enable you to distinguish between the following pair of chemicals. Describe what happens with each chemical or state 'no visible reaction'.
Sodium sulphate solution and sodium chloride solution.
उत्तर
Sodium sulphate solution and sodium chloride solution can be distinguished by using barium chloride solution. Barium chloride solution on being added to sodium sulphate solution forms a white precipitate which is insoluble in conc. HCl whereas sodium chloride shows no reaction with barium chloride solution.
APPEARS IN
संबंधित प्रश्न
The following are pertaining to the laboratory preparation of hydrogen chloride gas.
Write the equation for its preparation mentioning the condition required.
State your observation in given case When dilute hydrochloric acid is added to sodium carbonate crystals
For the preparation of hydrochloric acid in the laboratory:
Why is the direct absorption of hydrogen chloride gas in water not feasible?
Explain why hydrogen chloride gas is not collected over water.
Write the main difference in hydrogen chloride gas and hydrochloric acid.
The given set up in the figure is for the preparation of an acid.
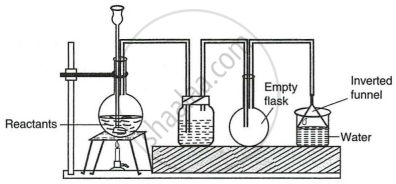
- Name the acid prepared by this method.
- Name the reactants used.
- Why an empty flask is used?
- What is the drying agent used? Why is this drying agent chosen?
- What is the role of the inverted funnel in the arrangement?
The drying agent used to dry \[\ce{HCl}\] gas is ______.
Draw a labelled diagram and explain the laboratory preparation of hydrogen chioride gas.
What are the important precautions?
Explain, why (or give reasons for)
Hydrogen chloride is not collected over water.
Describe an experiment to prove the following:
HCI gas contains the element chlorine.
Mention the reaction condition and give balanced equation to obtain: Cl2 gas from HCI acid.
Write the equation for :
The preparation of hydrogen chloride from sodium chloride and sulphuric acid. State whether the sulphuric acid should be concentrated or dilute.
State the observation for action of dilute hydrochloiric acid or iron (II) sulphate.
Answer the following question related to the laboratory preparation of the hydrogen chloride gas:
Why is sodium chloride preferred to other metallic chlorides?
In the laboratory preparation, HCl gas is dried by passing through ______.
Identify the gas evolved in the following reaction:
\[\ce{MnO2}\] reacts with concentrated \[\ce{HCl}\].
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCl ->}\]
