Advertisements
Advertisements
प्रश्न
When we heat an object, it expands. Is work done by the object in this process? Is heat given to the object equal to the increase in its internal energy?
उत्तर
When we heat an object, it expands, i.e. its volume increases.
Work done by the system, ΔW = P Δ V
Using the first law of thermodynamics, we get
ΔQ = ΔU + ΔW
Since the volume changes, ΔW has some non-zero positive value. Thus, heat given to the object is not equal to the increase in the internal energy of the system.
APPEARS IN
संबंधित प्रश्न
The first law of thermodynamics is a statement of ____________ .
Refer to figure. Let ∆U1 and ∆U2 be the change in internal energy in processes A and B respectively, ∆Q be the net heat given to the system in process A + B and ∆W be the net work done by the system in the process A + B.

(a) ∆U1 + ∆U2 = 0
(b) ∆U1 − ∆U2 = 0
(c) ∆Q − ∆W = 0
(d) ∆Q + ∆W = 0
The internal energy of an ideal gas decreases by the same amount as the work done by the system.
(a) The process must be adiabatic.
b) The process must be isothermal.
(c) The process must be isobaric.
(d) The temperature must decrease.
Calculate the change in internal energy of a gas kept in a rigid container when 100 J of heat is supplied to it.
The internal energy of a gas is given by U = 1.5 pV. It expands from 100 cm3 to 200 cm3against a constant pressure of 1.0 × 105 Pa. Calculate the heat absorbed by the gas in the process.
Consider the cyclic process ABCA, shown in figure, performed on a sample of 2.0 mol of an ideal gas. A total of 1200 J of heat is withdrawn from the sample in the process. Find the work done by the gas during the part BC.
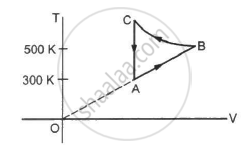
Figure shows the variation in the internal energy U with the volume V of 2.0 mol of an ideal gas in a cyclic process abcda. The temperatures of the gas at b and c are 500 K and 300 K respectively. Calculate the heat absorbed by the gas during the process.

Find the change in the internal energy of 2 kg of water as it is heated from 0°C to 4°C. The specific heat capacity of water is 4200 J kg−1 K−1 and its densities at 0°C and 4°C are 999.9 kg m−3 and 1000 kg m−3 respectively. Atmospheric pressure = 105 Pa.
ΔU is equal to ____________ work.
Which of the following are TRUE for a reversible isothermal process?
(i) ∆U = 0
(ii) ∆H = 0
(iii) Q = W
(iv) ∆T = 0
In a given process for an ideal gas, dW = 0 and dQ < 0. Then for the gas ____________.
Change in internal energy, when 4 KJ of work is done on the system and 1 KJ heat is given out by the system, is:
Is it possible to increase the temperature of a gas without adding heat to it? Explain.
Write the mathematical equation for the first law of thermodynamics for:
Isothermal process
Which among the following equations represents the first law of thermodynamics under isobaric conditions?
The amount of work done in increasing the voltage across the plates of capacitor from 5 V to 10 V is W. The work done in increasing it from 10 V to 15 V will be ______.
An ideal gas (γ = 1.5) is expanded adiabatically. How many times has the gas had to be expanded to reduce the root mean square velocity of molecules two times?
A given system undergoes a change in which the work done by the system equals the decrease in its internal energy. The system must have undergone an ______.
Explain the formulation of first law of thrmodynamics.
Define the isothermal process.
