Advertisements
Advertisements
Question
A student takes 2 mL acetic acid in a dry test tube and adds a pinch of sodium hydrogen carbonate to it. He makes the following observations:
I. A colourless and odourless gas evolves with a brisk effervescence.
II. The gas turns lime water milky when passed through it.
III. The gas burns with an explosion when a burning splinter is brought near it.
IV. The gas extinguishes the burning splinter that is brought near it.
The correct observations are:
(A) I, II, and III
(B) II, III and IV
(C) III, IV and I
(D) IV, I and II
Solution
(D)
When 2 mL acetic acid is taken in a dry test tube and a pinch of sodium hydrogen carbonate is added to it, a colourless and odourless gas evolves with a brisk effervescence which is CO2.
When CO2 is passed through lime water it turns lime water milky. 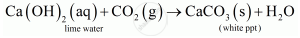
CO2 also extinguishes the burning splinter when it is brought near it.
APPEARS IN
RELATED QUESTIONS
Which of the following will give brisk effervescence with sodium hydrogen carbonate and why?
CH3COOH, CH3CH2OH
Why is the conversion of ethanol into ethanoic acid an oxidation reaction?
What happens when propanoic acid is warmed with methanol in the presence of a few drops of concentrated sulphuric acid? Write equation of the reaction involved.
What do you observe when acetic acid is added to ethyl alcohol in the presence of sulphuric acid?
While studying saponification reactions, the following comments were noted down by the students :
(I) Soap is a salt of fatty acids.
(II) The reaction mixture is basic in nature.
(III) In this reaction heat is absorbed.
(IV) This reaction is not a neutralisation reaction.
Which of these are the correct comments ?
(A) I and III only
(B) I, II and III
(C) II, III and IV
(D) I and II only
Under what conditions does ethane get converted to ethyl alcohol?
Write the important uses of acetic acid.
What is thr boilng point of acetic acid?
Draw the structural formula for the following:
Ethanoic acid
Two statements are given - one labelled Assertion (A) and the other labelled Reason (R).
Assertion (A): Esterification is a process in which a sweet-smelling substance is produced.
Reason (R): When esters react with sodium hydroxide an alcohol and sodium salt of carboxylic acid are obtained.
