Advertisements
Advertisements
Question
Activation energy of a chemical reaction can be determined by ______.
Options
determining the rate constant at standard temperature.
determining the rate constants at two temperatures.
determining probability of collision.
using catalyst.
Solution
Activation energy of a chemical reaction can be determined by determining the rate constants at two temperatures.
Explanation:
Where, Ea = activation energy
T2 = higher temperature
T1 = lower temperature
k1 = rate constant at temperature T1
k2 = rate constant at temperature T2
This equation is known as Arrhenius equation.
APPEARS IN
RELATED QUESTIONS
Consider a certain reaction
The decomposition of A into product has value of k as 4.5 × 103 s−1 at 10°C and energy of activation 60 kJ mol−1. At what temperature would k be 1.5 × 104 s−1?
The rate constant of a first order reaction are 0.58 S-1 at 313 K and 0.045 S-1 at 293 K. What is the energy of activation for the reaction?
Explain the following terms :
Half life period of a reaction (t1/2)
The decomposition of a hydrocarbon has value of rate constant as 2.5×104s-1 At 27° what temperature would rate constant be 7.5×104 × 3 s-1if energy of activation is 19.147 × 103 J mol-1 ?
Consider figure and mark the correct option.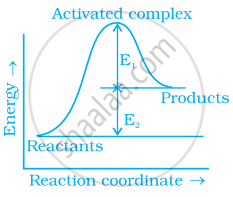
Which of the following statements are in accordance with the Arrhenius equation?
(i) Rate of a reaction increases with increase in temperature.
(ii) Rate of a reaction increases with decrease in activation energy.
(iii) Rate constant decreases exponentially with increase in temperature.
(iv) Rate of reaction decreases with decrease in activation energy.
The equation k =
An exothermic reaction X → Y has an activation energy 30 kJ mol-1. If energy change ΔE during the reaction is - 20 kJ, then the activation energy for the reverse reaction in kJ is ______.
A schematic plot of ln Keq versus inverse of temperature for a reaction is shown below
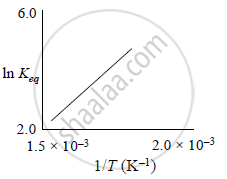
The reaction must be:
