Advertisements
Advertisements
Question
Consider a certain reaction \[\ce{A -> Products}\] with k = 2.0 × 10−2 s−1. Calculate the concentration of A remaining after 100 s if the initial concentration of A is 1.0 mol L−1.
Solution
k = 2.0 × 10−2 s−1, T1 = 0, [A]1 = 1.0 M,
T2 = 100 s, [A]2 = ?
k = `1/("T"_2 - "T"_1) ln (["A"]_1)/(["A"]_2)`
2.0 × 10−2 = `1/(100 - 0) ln 1.0/(["A"]_2)`
(2.0 × 10−2) × 100 = `ln 1.0/(["A"]_2)`
2 = `ln 1/(["A"]_2)`
7.389 = `1/(["A"]_2)`
∴ [A]2 = `1/7.389`
= 0.135 mol L−1
APPEARS IN
RELATED QUESTIONS
Consider the reaction
`3I_((aq))^-) +S_2O_8^(2-)->I_(3(aq))^-) + 2S_2O_4^(2-)`
At particular time t, `(d[SO_4^(2-)])/dt=2.2xx10^(-2)"M/s"`
What are the values of the following at the same time?
a. `-(d[I^-])/dt`
b. `-(d[S_2O_8^(2-)])/dt`
c. `-(d[I_3^-])/dt`
The rate constant of a first order reaction increases from 4 × 10−2 to 8 × 10−2 when the temperature changes from 27°C to 37°C. Calculate the energy of activation (Ea). (log 2 = 0.301, log 3 = 0.4771, log 4 = 0.6021)
What will be the effect of temperature on rate constant?
The decomposition of hydrocarbon follows the equation k = `(4.5 xx 10^11 "s"^-1) "e"^(-28000 "K"//"T")`
Calculate Ea.
The rate of a reaction quadruples when the temperature changes from 293 K to 313 K. Calculate the energy of activation of the reaction assuming that it does not change with temperature.
In the Arrhenius equation for a first order reaction, the values of ‘A’ of ‘Ea’ are 4 x 1013 sec-1 and 98.6 kJ mol-1 respectively. At what temperature will its half life period be 10 minutes?
[R = 8.314 J K-1 mol-1]
The decomposition of a hydrocarbon has value of rate constant as 2.5×104s-1 At 27° what temperature would rate constant be 7.5×104 × 3 s-1if energy of activation is 19.147 × 103 J mol-1 ?
Write a condition under which a bimolecular reaction is kinetically first order. Give an example of such a reaction. (Given : log2 = 0.3010,log 3 = 0.4771, log5 = 0.6990).
The chemical reaction in which reactants require high amount of activation energy are generally ____________.
Activation energy of a chemical reaction can be determined by ______.
Which of the following graphs represents exothermic reaction?
(a)
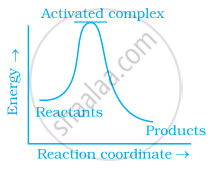
(b)
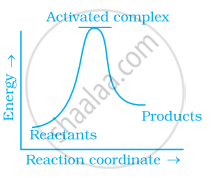
(c)
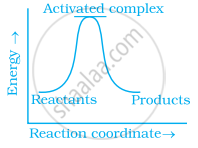
Which of the following statements are in accordance with the Arrhenius equation?
(i) Rate of a reaction increases with increase in temperature.
(ii) Rate of a reaction increases with decrease in activation energy.
(iii) Rate constant decreases exponentially with increase in temperature.
(iv) Rate of reaction decreases with decrease in activation energy.
Why does the rate of a reaction increase with rise in temperature?
Thermodynamic feasibility of the reaction alone cannot decide the rate of the reaction. Explain with the help of one example.
What happens to most probable kinetic energy and the energy of activation with increase in temperature?
The activation energy in a chemical reaction is defined as ______.
An exothermic reaction X → Y has an activation energy 30 kJ mol-1. If energy change ΔE during the reaction is - 20 kJ, then the activation energy for the reverse reaction in kJ is ______.
What happens to the rate constant k and activation energy Ea as the temperature of a chemical reaction is increased? Justify.
