Advertisements
Advertisements
Question
An equilibrium constant of 3.2 × 10-6 for a reaction means, the equilibrium is
Options
largely towards forward direction
largely towards reverse direction
never established
none of these
Solution
largely towards reverse direction
APPEARS IN
RELATED QUESTIONS
\[\ce{[CO(H2O)6]^2+ (aq) (pink) + 4Cl- (aq) <=> [CoCl4]^2- (aq) (blue) + 6 H2O (l)}\]
In the above reaction at equilibrium, the reaction mixture is blue in colour at room temperature. On cooling this mixture, it becomes pink in color. On the basis of this information, which one of the following is true?
What is the relation between Kp and Kc? Given one example for which Kp is equal to Kc.
When the numerical value of the reaction quotient (Q) is greater than the equilibrium constant, in which direction does the reaction proceed to reach equilibrium?
For the reaction, \[\ce{A2(g) + B2(g) <=> 2AB(g); \Delta H}\] is -ve.
the following molecular scenes represent differenr reaction mixture. (A-green, B-blue)
| Closed ← |
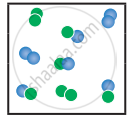 |
 |
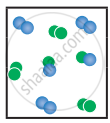 |
| System | At equilibrium | (x) | (y) |
- Calculate the equilibrium constant Kp and (Kc).
- For the reaction mixture represented by scene (x), (y) the reaction proceed in which directions?
- What is the effect of an increase in pressure for the mixture at equilibrium?
Write the balanced chemical equation for an equilibrium reaction for which the equilibrium constant is given by expression.
`"K"_"C" = (["NH"_3]^4["O"_2]^5)/(["NO"]^4["H"_2"O"]^6)`
What is the effect of added Inert gas on the reaction at equilibrium?
1 mol of CH4, 1 mole of CS2 and 2 mol of H2S are 2 mol of H2 are mixed in a 500 ml flask. The equilibrium constant for the reaction Kc = 4 x 10-2 mol2 lit-2. In which direction will the reaction proceed to reach equilibrium?
At particular temperature Kc = 4 × 10-2 for the reaction, \[\ce{H2S (g) <=> H2(g) +1/2 S2(g)}\]. Calculate the Kc for the following reaction.
\[\ce{3H2S (g) <=> 3H2 (g) + 3/2 S2 (g)}\]
The partial pressure of carbon dioxide in the reaction
\[\ce{CaCO3(s) <=> CaO(s) + CO2(g)}\] is 1.017 × 10-3 atm at 500°C. Calculate Kp at 600°C for the reaction. H for the reaction is 181 KJ mol-1 and does not change in the given range of temperature.
The equilibrium constant Kp for the reaction \[\ce{N2 (g) + 3H2 (g) <=> 2NH3 (g)}\] is 8.19 × 102 at 298 K and 4.6 × 10-1 at 498 K. Calculate ∆H° for the reaction.
