Advertisements
Advertisements
Question
Explain with the help of suitable example polar covalent bond.
Solution 1
When two dissimilar atoms having different electronegativities combine to form a covalent bond, the bond pair of electrons is not shared equally. The bond pair shifts towards the nucleus of the atom having greater electronegativity. As a result, electron distribution gets distorted and the electron cloud is displaced towards the electronegative atom.
As a result, the electronegative atom becomes slightly negatively charged while the other atom becomes slightly positively charged. Thus, opposite poles are developed in the molecule and this type of a bond is called a polar covalent bond.
HCl, for example, contains a polar covalent bond. Chlorine atom is more electronegative than hydrogen atom. Hence, the bond pair lies towards chlorine and therefore, it acquires a partial negative charge.
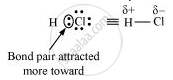
Solution 2
When two atoms with different electronegativity are linked to each other by covalent bond, the shared electron pair will not in the centre because of the difference in electronegativity. For example, in hydrogen flouride molecule, fluoride has greater electronegativity than hydrogen. Thus, the shared electron pair is displaced more towards ’fluorine atom, the later will acquire a partial negative charge (∂–). At the same time hydrogen atom will have a partial positive charge (∂+). Such a covalent bond is known as polar covalent bond or simply polar bond. It is represented as
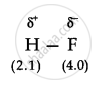
APPEARS IN
RELATED QUESTIONS
Although both CO2 and H2O are triatomic molecules, the shape of H2O molecule is bent while that of CO2 is linear. Explain this on the basis of dipole moment.
Write the significance/applications of dipole moment.
Define electronegativity.
How does electronegativity differ from electron gain enthalpy?
Arrange the bonds in order of increasing ionic character in the molecules: LiF, K2O, N2, SO2 and ClF3.
Which out of NH3 and NF3 has higher dipole moment and why?
Polarity in a molecule and hence the dipole moment depends primarily on electronegativity of the constituent atoms and shape of a molecule. Which of the following has the highest dipole moment?
Arrange the following bonds in order of increasing ionic character giving reason.
\[\ce{N - H, F - H, C - H}\] and \[\ce{O - H}\]
Which of the following is CORRECTLY matched?
If AB4 molecule is a polar molecule, a possible geometry of AB4 is ______.
Dipole moment of HX is 2.59 × 10−30 coulomb-metre. Bond length of HX is 1.39 Å. The ionic character of molecule is ______%.
The dipole moments of the given molecules are such that ______.
The observed dipole-moment of HCl molecule is 1.03 D. If \[\ce{H - Cl}\] bond distance is 1.275 Å and electronic charge is 4.8 × 10-10 esu. What is the percent polarity of HCl?
