Advertisements
Advertisements
Question
Write the significance/applications of dipole moment.
Solution 1
In heteronuclear molecules, polarization arises due to a difference in the electronegativities of the constituents of atoms. As a result, one end of the molecule acquires a positive charge while the other end becomes negative. Hence, a molecule is said to possess a dipole.
The product of the magnitude of the charge and the distance between the centres of positive-negative charges is called the dipole moment (μ) of the molecule. It is a vector quantity and is represented by an arrow with its tail at the positive centre and head pointing towards a negative centre.
Dipole moment (μ) = charge (Q) × distance of separation (r)
The SI unit of a dipole moment is ‘esu’.
1 esu = 3.335 × 10–30 Cm
Dipole moment is the measure of the polarity of a bond. It is used to differentiate between polar and non-polar bonds since all non-polar molecules (e.g. H2, O2) have zero dipole moments. It is also helpful in calculating the percentage ionic character of a molecule.
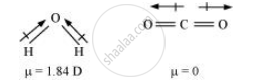
Solution 2
In predicting the nature of the molecules: Molecules with specific dipole moments are polar in nature and those of zero dipole moments are non-polar in nature.
In the determination of shapes of molecules.
In calculating the percentage ionic character.
APPEARS IN
RELATED QUESTIONS
Although both CO2 and H2O are triatomic molecules, the shape of H2O molecule is bent while that of CO2 is linear. Explain this on the basis of dipole moment.
Define electronegativity.
How does electronegativity differ from electron gain enthalpy?
Explain with the help of suitable example polar covalent bond.
Arrange the bonds in order of increasing ionic character in the molecules: LiF, K2O, N2, SO2 and ClF3.
Which out of NH3 and NF3 has higher dipole moment and why?
Polarity in a molecule and hence the dipole moment depends primarily on electronegativity of the constituent atoms and shape of a molecule. Which of the following has the highest dipole moment?
Which of the following is CORRECTLY matched?
If AB4 molecule is a polar molecule, a possible geometry of AB4 is ______.
Dipole moment of HX is 2.59 × 10−30 coulomb-metre. Bond length of HX is 1.39 Å. The ionic character of molecule is ______%.
The dipole moments of the given molecules are such that ______.
The observed dipole-moment of HCl molecule is 1.03 D. If \[\ce{H - Cl}\] bond distance is 1.275 Å and electronic charge is 4.8 × 10-10 esu. What is the percent polarity of HCl?
The dipole moments of CCl4, CHCl3 and CH4 are in the order:
