Advertisements
Advertisements
Question
Find the radius and energy of a He+ ion in the states (a) n = 1, (b) n = 4 and (c) n = 10.
Solution
For He+ ion,
Atomic number, Z = 2
For hydrogen like ions, radius (r) of the nth state is given by
`r = (0.53 n^2)/ZÅ`
Here
Z= Atomic number of ions
n = Quantum number of the state
Energy (E) of the nth state is given by
`E_n = -(13.6 Z^2)/(n^2)`
(a)
for n = 1
Radius,
`r = (0.53xx(1)^2)/2`
`Å = 0.265 A^0`
Energy, En = `(-13.6 xx 4)/1`
= - 54.4 ev
(b)
For n = 4,
Radius, `r = (0.53xx16)/2 = 4.24 Å `
Energy, `E = (-13.6xx4)/16= -3.4 eV`
(c)
For n = 10,
`Radius, r = (0.53xx100)/2`
= 26.5 Å
Energy, E = `(-13.6 xx 4 )/100 = -0.544 eV`
APPEARS IN
RELATED QUESTIONS
Which wavelengths will be emitted by a sample of atomic hydrogen gas (in ground state) if electrons of energy 12.2 eV collide with the atoms of the gas?
What will be the energy corresponding to the first excited state of a hydrogen atom if the potential energy of the atom is taken to be 10 eV when the electron is widely separated from the proton? Can we still write En = E1/n2, or rn = a0 n2?
In which of the following transitions will the wavelength be minimum?
Which of the following curves may represent the speed of the electron in a hydrogen atom as a function of trincipal quantum number n?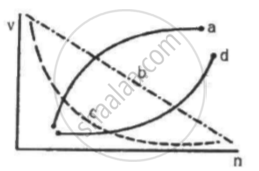
As one considers orbits with higher values of n in a hydrogen atom, the electric potential energy of the atom
A hydrogen atom in ground state absorbs 10.2 eV of energy. The orbital angular momentum of the electron is increased by
Find the binding energy of a hydrogen atom in the state n = 2.
(a) Find the first excitation potential of He+ ion. (b) Find the ionization potential of Li++ion.
A hydrogen atom in state n = 6 makes two successive transitions and reaches the ground state. In the first transition a photon of 1.13 eV is emitted. (a) Find the energy of the photon emitted in the second transition (b) What is the value of n in the intermediate state?
A gas of hydrogen-like ions is prepared in a particular excited state A. It emits photons having wavelength equal to the wavelength of the first line of the Lyman series together with photons of five other wavelengths. Identify the gas and find the principal quantum number of the state A.
Find the maximum angular speed of the electron of a hydrogen atom in a stationary orbit.
Suppose, in certain conditions only those transitions are allowed to hydrogen atoms in which the principal quantum number n changes by 2. (a) Find the smallest wavelength emitted by hydrogen. (b) List the wavelength emitted by hydrogen in the visible range (380 nm to 780 nm).
The average kinetic energy of molecules in a gas at temperature T is 1.5 kT. Find the temperature at which the average kinetic energy of the molecules of hydrogen equals the binding energy of its atoms. Will hydrogen remain in molecular from at this temperature? Take k = 8.62 × 10−5 eV K−1.
Show that the ratio of the magnetic dipole moment to the angular momentum (l = mvr) is a universal constant for hydrogen-like atoms and ions. Find its value.
Electrons are emitted from an electron gun at almost zero velocity and are accelerated by an electric field E through a distance of 1.0 m. The electrons are now scattered by an atomic hydrogen sample in ground state. What should be the minimum value of E so that red light of wavelength 656.3 nm may be emitted by the hydrogen?
Consider an excited hydrogen atom in state n moving with a velocity υ(ν<<c). It emits a photon in the direction of its motion and changes its state to a lower state m. Apply momentum and energy conservation principles to calculate the frequency ν of the emitted radiation. Compare this with the frequency ν0 emitted if the atom were at rest.
In a hydrogen atom the electron moves in an orbit of radius 0.5 A° making 10 revolutions per second, the magnetic moment associated with the orbital motion of the electron will be ______.
Positronium is just like a H-atom with the proton replaced by the positively charged anti-particle of the electron (called the positron which is as massive as the electron). What would be the ground state energy of positronium?
