Advertisements
Advertisements
Question
Give one example of a decomposition reaction which is carried out with electricity.
Solution
Example of the decomposition reaction are as follows:
with electricity:
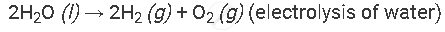
APPEARS IN
RELATED QUESTIONS
What type of reaction is represented by the following equation?
NH4 CI → NH3 + HCI
What type of chemical reaction is represented by the following equation?
X → Y + Z
Name the product formed on strongly heating ferrous sulphate crystals. What type of chemical reaction occurs in this change?
Which of the following can be decomposed by the action of light?
(a) NaCl
(b) KCl
(c) AgCl
(d) CuCl
Answer the following question.
2 g of silver chloride is taken in a china dish and the china dish is placed in sunlight for some time. What will be your observation in this case? Write the chemical reaction involved in the form of a balanced chemical equation. Identify the type of chemical reaction.
Give a balanced equation for the following type of reaction:
A thermal decomposition reaction in which a compound decomposes to give two new compounds.
Differentiate between the following:
Electrolytic decomposition and photochemical decomposition.
Photolysis is a decomposition reaction caused by ______
Electrolysis of water is a decomposition reaction. The mole ratio of hydrogen and oxygen gases liberated during electrolysis of water is:
What is observed when silver chloride is exposed to sunlight? Give the type of reaction involved.
