Advertisements
Advertisements
Question
Give the structure of A, B and C in the following reaction:
\[\ce{CH3CH2I ->[NaCN] A ->[OH-][Partial hydrolysis] B ->[NaOH + Br2] C}\]
Solution
\[\begin{array}{cc}
\phantom{.......................}\ce{O}\\
\phantom{.......................}||\\
\ce{\underset{Iodoethane}{CH3CH2I} ->[NaCN] \underset{(A)}{\underset{Propane nitrile}{CH3CH2CN}} ->[OH-][Partial hydrolysis] \underset{(B)}{\underset{Propanamide}{CH3CH2-C-NH2}} ->[NaOH + Br2] \underset{(C)}{\underset{Ethanamine}{CH3CH2-NH2}}}
\end{array}\]
APPEARS IN
RELATED QUESTIONS
Give the structures of A, B and C in the following reaction:
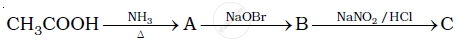
Write the reactions of aromatic with nitrous acid.
Give the structures of A, B and C in the following reactions :
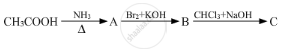
Which of the following amines exhibits maximum degree of intermolecular hydrogen bonding?
What is the molar mass of the amine formed when acetamide undergoes Hofmann bromamide degradation?
The reduction of alkyl cyanide with sodium and ethanol to give primary amines is, ____________.
Quaternary ammonium salt is formed:
Given below are two statements labelled as Assertion (A) and Reason (R).
Assertion (A): Alkyl halides are insoluble in water.
Reason (R): Alkyl halides have halogen attached to sp3 hybrid carbon.
Select the most appropriate answer from the options given below:
The source of nitrogen in Gabriel synthesis of amines is ______.
Among the following amines, the strongest Brönsted base is:
Describe Gabriel's phthalimide synthesis. (Give reaction)
Ethylamine can be prepared by the action of bromine and caustic potash on which compound?
Acetamide and ethyl amide can be distinguished by reacting with.
C6H5CONHCH3 can be converted into C6H5CH2NHCH3 by:-
A compound 'A' on reduction with iron scrap and hydrochloric acid gives compound 'B' with molecular formula C6H7N. Compound 'B' on reaction with CHCl3 and alcoholic KOH produces an obnoxious smell of carbylamine due to the formation of 'C'. Identify 'A', 'B' and 'C' and write the chemical reactions involved.
Which of the following amines can be prepared by Gabriel phthalimide reaction?
Identify the product ‘C’ in the following reaction.
\[\ce{Aniline ->[(CH3CH)2O][Pyridine] A ->[Br2][CH3COOH] B ->[H^+ or OH^-] C}\]
Which of the following would not be a good choice for reducing nitrobenzene to aniline?
Identify the compo ds A and B in the following reactions:
\[\ce{A ->[Nitrating mixture] B ->[(i) Sn/cone. HCI][(ii) NaOH] Aniline}\]
