Advertisements
Advertisements
Question
Which is a stronger reducing agent, Cr2+ or Fe2+ and why?
Solution
Cr2+ is a stronger reducing agent than Fe2+. In the formation of Cr2+ to Cr3+, the change is d4 → d3, but in the formation of Fe2+ to Fe3+, the change is d6 → d5. In a medium like water, d3 is more stable than d5.
APPEARS IN
RELATED QUESTIONS
In 3d series (Sc to Zn), which element has the lowest enthalpy of atomisation and why?
In the series Sc (Z = 21) to Zn (Z = 30), the enthalpy of atomization of zinc is the lowest, i.e., 126 kJ mol−1. Why?
NF3 is possible, but NF5 is not. Why?
Write the formula of an oxo-anion of Chromium (Cr) in which it shows the oxidation state equal to its group number
How would you account for the following?
Transition metals and their compounds act as catalysts.
The paramagnetic character in-3d-transition series elements increases-: up to Mn and then decreases.
Which of the following will not act as oxidising agents?
(i) \[\ce{CrO3}\]
(ii) \[\ce{MoO3}\]
(iii) \[\ce{WO3}\]
(iv) \[\ce{CrO^{2-}4}\]
Match the properties given in Column I with the metals given in Column II.
| Column I (Property) | Column II (Metal) | |
| (i) | Element with highest second ionisation enthalpy |
(a) \[\ce{Co}\] |
| (ii) | Element with highest third ionisation enthalpy |
(b) \[\ce{Cr}\] |
| (iii) | \[\ce{M}\] in \[\ce{M(CO)6}\] is | (c) \[\ce{Cu}\] |
| (iv) | Element with highest heat of atomisation |
(d) \[\ce{Zn}\] |
| (e) \[\ce{Ni}\] |
Assertion: The highest oxidation state of osmium is +8.
Reason: Osmium is a 5d-block element.
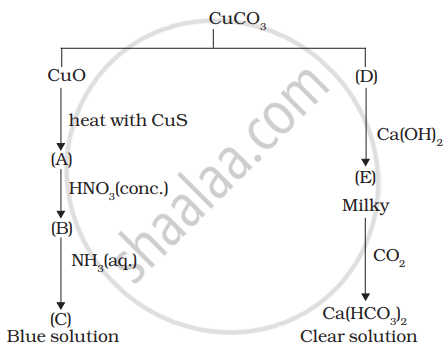
Identify A to E and also explain the reactions involved.
Which of the following maxm magnetic moment?
The product of oxidation of I– with \[\ce{MnO^{-}4}\] in alkaline medium is:-
Passing H2S gas into a mixture of Mn2+ and Ni2+, Cu2+, ions in an acidified aqueous solution precipitates.
The complex showing a spin-span magnetic moment of 2.82 B.M. is :-
The number of terminal oxygen atoms present in the product B obtained from the following reactions is:
\[\ce{FeCr2O4 + Na2CO3 + O2 -> A + Fe2O3 + CO2}\]
\[\ce{A + H^+ -> B + H2O + Na^+}\]
Write the number of unpaired electrons in Cr3+.
(Atomic number of Cr = 24)
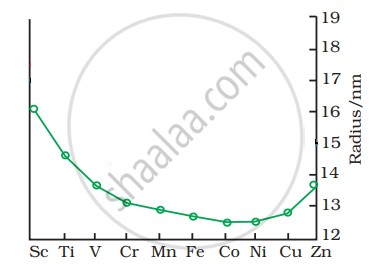
The trend of which property is represented by the following graph?
Explain the magnetic properties of d-block (or transition) elements.
The compounds of \[\ce{Ti^4+}\] ions are colourless due to ______.
Give a reason for the following.
Some transition metals and their compounds get attracted towards the magnetic field.
