Advertisements
Advertisements
Question
With a labelled diagram describe in brief an activity to show the formation of ester.
Solution
The activity given below shows the formation of ester :
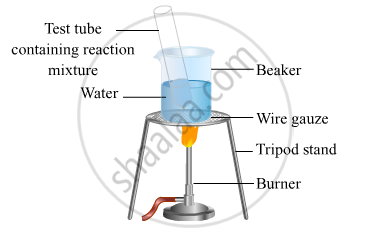
- Take 1 mL ethanol (absolute alcohol) and 1 mL glacial acetic acid along with a few drops of concentrated sulphuric acid in a test tube.
- Warm in a water-bath for at least five minutes as shown in the figure below.
- Pour into a beaker containing 20-50 mL of water.
- After some time, there will be the formation of a fruity smell compound. The compound formed is an ester.
The formation of the ester can be represented by the chemical reaction as:
\[\ce{\underset{\text{(Ethanoic acid)}}{CH3COOH} + \underset{\text{(Ethanol)}}{C2H5OH}->[conc. H2SO4] \underset{\text{(Ester)}}{CH3COOC2H5} + H2O}\]
APPEARS IN
RELATED QUESTIONS
Write three different chemical reactions showing the conversion of ethanoic acid to sodium ethanoate. Write balanced chemical equation in each case. Write the name of the reactants and the products other ethanoic acid and sodium ethanoate in each case.
Fill in the blanks from the choices given within brackets:
The basicity of acetic acid is-------- (3, 1, 4).
What happens when ethanoic acid reacts with sodium carbonate? Write chemical equation of the reaction involved.
Name the functional group present in the following compound:
HCOOH
How is ethyne prepared in the laboratory?
How will you carry out the following conversions?
Ethene to acetic acid
Write a balanced chemical equation for the following:
A mixture of sodalime and sodium acetate is heated.
CH3–CH2–CHO : propanal : : CH3–COOH : _______
Explain the following reaction with an example.
Esterification
A student while observing the properties of acetic acid would report that this smells like ______.
