Advertisements
Advertisements
Questions
Write a commercial method for preparation of glucose.
How is glucose prepared on a commercial scale?
Solution
Commercial method for preparation of glucose: Commercially glucose is obtained by hydrolysis of starch by boiling it with dilute sulphuric acid at 393K under 2 to 3 atm pressure.
\[\ce{\underset{\text{Starch}}{(C6H10O5)}_{{n}} + {n}H2O ->[H+][393K, 2-3 atm] \underset{\text{Glucose}}{{n}C6H12O6}}\]
APPEARS IN
RELATED QUESTIONS
Draw a neat diagram for the following:
Haworth formula of maltose
Draw the structure of α-D glucopyranose.
Explain the preparation of glucose from sucrose.
Draw the Haworth projection structure of the following.
α-D-(–)-Fructofuranose
Write a chemical reaction to convert glucose into glucose cyanohydrin.
What is monosaccharide?
Identify the given structure 'P' and 'Q'.
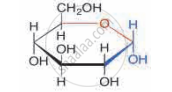
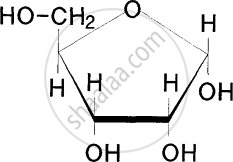
Which of the following carbohydrate is sweet to taste?
Carbohydrates can contain which of the following chemical groups?
From the following identify the group that is exclusively consists of polysaccharides.
From the following which is a heteropolysaccharide?
Identify the sugar having the molecular formula C6H1206.
Which reagent among the following is used to confirm presence of aldehydic carbonyl group in glucose?
All these carbohydrates contain \[\ce{1 -> 4β}\] glycosidic linkage, EXCEPT ____________.
Which one of the following is a carbohydrate but does NOT follow the general formula of carbohydrate?
Which one of the following is generally applicable to polysaccharides?
Which of the following statements is NOT true for glyceraldehyde?
Glucose and gluconic acid treated with dilute nitric acid forms saccharic acid. What does this indicate?
How many hydroxyl groups are present in Erythrulose?
How many optical isomers are possible for a compound having four asymmetric carbon atoms?
Which one of the following rotates the plane polarized light towards left?
Complete hydrolysis of cellulose gives ____________.
What are reducing and non-reducing sugars?
Glucose on oxidation with bromine water yields gluconic acid. This reaction confirms the presence of ______.
Hydrolysis of sucrose is called ______.
Which carbon atoms of fructose are bonded together through oxygen forming fructofuranose?
Match the Column I with Column II and choose the correct answer from options below:
| Column I | Column II |
| A. Purine | 1. Glycogen |
| B. Pyrimidine | 2. Cellulose |
| C. Structural polysaccharide | 3. Glucagon |
| D. Storage polysaccharide | 4. Adenine |
| 5. Cytosine |
Identify the monosaccharide containing only one asymmetric carbon atom in its molecule.
The glycosidic linkage present in maltose is ______.
\[\ce{CH2OH-CO-(CHOH)4-CH2OH}\] is an example of ______.
