SSC (English Medium)
SSC (Marathi Semi-English)
Academic Year: 2018-2019
Date: March 2019
Advertisements
When a liquid is getting converted into solid, the latent heat is ………………………………
Chapter: [0.05] Heat
Identify the type of following reaction :
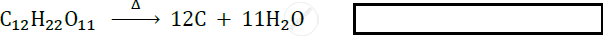
Chapter: [0.03] Chemical Reactions and Equations [0.12] The Magic of Chemical Reactions
In Dobereiner’s triad containing Li, Na, K, if atomic masses of Lithium & Potassium are 6.9 and 39.1, then what will be the atomic mass of Sodium?
Chapter: [0.02] Periodic Classification of Elements [0.11] School of Elements
Considering first correlation, complete the second.
Hubble telescope : At 569 km above the earth’s surface
Orbit of Hubble telescope : .............................
Chapter: [0.1] Space Missions
Observe the figure and write accurate conclusion regarding refraction of light.
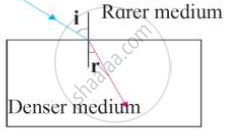
Chapter: [0.17] Wonders of Light 2 [0.17] Wonders of Light 2
Write the correct option by observing the figures.
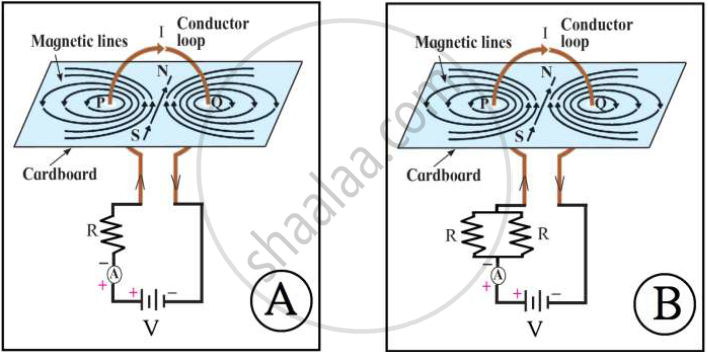
Magnetic field in A is stronger.
Magnetic field in B is stronger.
Magnetic fields in A and B are same.
Magnetic fields in A and B are weaker.
Chapter: [0.04] Effects of Electric Current
A student obtained clear image of window grills on the screen. But the teacher told him to get the image of a tree far away, instead of window. To get a clear image, the lens must be ............................
moved towards the screen.
moved away from the screen.
moved behind the screen.
moved far away from the screen.
Chapter: [0.07] Lenses
Advertisements
What would be the correct order if Zn, Fe, Al and Cu are arranged in increasing order of reactivity?
Cu, Fe, Zn, Al
Al, Cu, Fe, Zn
Zn, Al, Cu, Fe
Fe, Zn, Al, Cu.
Chapter: [0.02] Periodic Classification of Elements [0.11] School of Elements
The following structural formula belongs to which carbon compound?

Camphor
Benzene
Starch
Glucose
Chapter:
What will be the weight of a person on earth, who weighs 9N on the moon?
3 N
15 N
45 N
54 N
Chapter: [0.01] Gravitation
Statement 1 : Electric current ( flow of electrons ) creates heat in the resistor.
Statement 2 : Heat in the resistor is created according to the rule of energy conservation.
Explain Statement 1 with the help of Statement 2.
Chapter: [0.04] Effects of Electric Current [0.14] The Electric Spark
Swarali has got the following observations while doing an experiment. Answer her questions with the help of observations.
Swarali observed that the light bent away from the normal, while travelling from dense medium to rarer medium. When Swarali increased the values of angle of incidence (i), the values of angle of refraction (r) went on increasing. But at a certain angle of incidence, the light rays returned into the dense medium.
So, Swarali has some questions. Answer them –
a) Name this certain value of ‘i ’. What is the value of ‘r’ at that time ?
b) Name this process of returning of light in dense medium. Explain the process.
Chapter:
What is the correct reason for blinking / flickering of stars? Explain it.
a) The blasts in the stars.
b) Absorption of star light by the atmosphere.
c) Motion of the stars.
d) Changing refractive index of gases in the atmosphere.
Chapter: [0.17] Wonders of Light 2 [0.17] Wonders of Light 2
Atomic number of metal ‚A‛ is 11, while atomic number of metal ‚B‛ is 20. Which of them will be more reactive? Write the chemical reaction of dilute HCl with metal ‚A‛.
Chapter: [0.02] Periodic Classification of Elements [0.11] School of Elements
Draw electron - dot structure and structural formula of methane.
Chapter:
Solve the problem.
If the mass of a planet is eight times the mass of the Earth and its radius is twice the radius of the Earth, what will be the escape velocity for that planet?
Chapter: [0.01] Gravitation
Advertisements
What are space expeditions? Explain their need and importance in your words.
Chapter: [0.1] Space Missions
A ball falls off a table and reaches the ground in 1 s. Assuming g=10 m/s2, calculate its speed on reaching the ground and the height of the table.
Chapter: [0.01] Gravitation
Write down what you understood from the following chemical reaction.
AgNO3(aq) + NaCI(aq) → AgCI ↓ + NaNO3(aq)
Chapter: [0.03] Chemical Reactions and Equations [0.12] The Magic of Chemical Reactions
Observe the straight chain hydrocarbons given below and answer the following
questions :
i) Which of the straight chain compounds from A and B is saturated and unsaturated straight chains?
ii) Name these straight chains.
iii) Write their chemical formulae and number of – CH2 - units.

Chapter:
i) Which principle is explained in this figure?
ii) Which rule is used to find out the direction of a force in this principle?
iii) In which machine this principle is used? Draw a diagram showing working of that machine
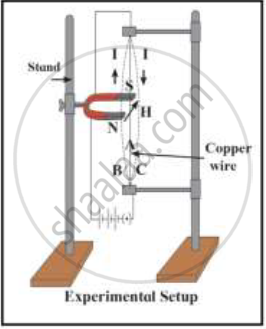
Chapter: [0.04] Effects of Electric Current [0.15] All about Electromagnetism
Study the following procedure and answer the questions below:
1. Take 3 spheres of iron, copper and lead of equal mass.
2. Put all the 3 spheres in boiling water in a beaker for some time.
3. Take 3 spheres out of the water. Put them immediately on a thick slab of wax.
4. Note, the depth that each sphere goes into the wax.
i) Which property of substance can be studied with this procedure?
ii) Describe that property in minimum words.
iii) Explain the rule of heat exchange with this property.
Chapter: [0.05] Heat
Complete the following flow chart and answer the questions below:
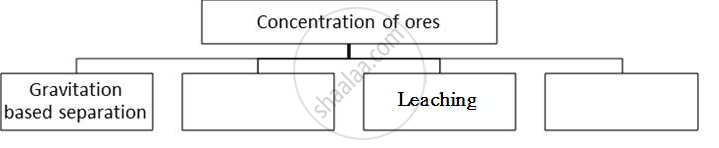
i) In which method pine oil is used?
ii) Explain what method of concentration in brief.
Chapter: [0.08] Metallurgy
Complete the process of iron rusting by filling the blanks. Suggest a way to prohibit the process.
The iron rust is formed due to........................... reaction. Different
regions on iron surface become anode and cathode.
Reaction on anode region :
`F_e(s) → Fe^(2+) (aq) +2e^-`
Reaction on anode region :
`O_2(g) + 4H^+(aq) +............................ → 2H_2 O (l) `
When Fe2+ ions migrate from anode region they react with ................... to form Fe3+ ions.
A reddish coloured hydrated oxide is formed from ............... ions. It is called rust.
`2Fe_(3+) (aq) + 4H_2O(l) → ................. + 6H_+(aq) `
A way to prevent rusting ..................................................................
Chapter: [0.03] Chemical Reactions and Equations [0.08] Metallurgy
An element has its electron configuration as 2,8,2. Now answer the following questions.
i. What is the atomic number of this element?
ii. What is the valency of this element?
iii. What is the group of this element?
iv. To which period does this element belong?
v. With which of the following elements would this element resemble?
`_7N , _4Be , _18Ar , _17CL`
Chapter: [0.02] Periodic Classification of Elements
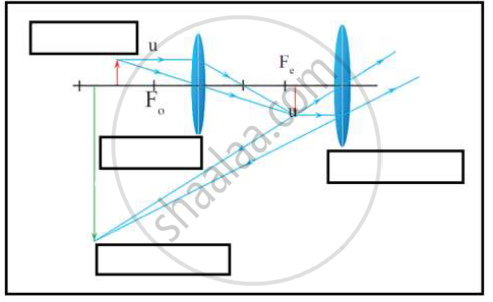
i. Which type of microscope has the arrangement of lenses shown in the adjoining figure?
ii. Label the figure correctly.
iii. Write the working of this microscope.
iv. Where does this microscope used?
v. Suggest a way to increase the efficiency of this microscope.
Chapter: [0.07] Lenses [0.16] Wonders of Light 1
Other Solutions
Submit Question Paper
Help us maintain new question papers on Shaalaa.com, so we can continue to help studentsonly jpg, png and pdf files
Maharashtra State Board previous year question papers 10th Standard Board Exam Science and Technology 1 with solutions 2018 - 2019
Previous year Question paper for Maharashtra State Board 10th Standard Board Exam -2019 is solved by experts. Solved question papers gives you the chance to check yourself after your mock test.
By referring the question paper Solutions for Science and Technology 1, you can scale your preparation level and work on your weak areas. It will also help the candidates in developing the time-management skills. Practice makes perfect, and there is no better way to practice than to attempt previous year question paper solutions of Maharashtra State Board 10th Standard Board Exam.
How Maharashtra State Board 10th Standard Board Exam Question Paper solutions Help Students ?
• Question paper solutions for Science and Technology 1 will helps students to prepare for exam.
• Question paper with answer will boost students confidence in exam time and also give you an idea About the important questions and topics to be prepared for the board exam.
• For finding solution of question papers no need to refer so multiple sources like textbook or guides.
