Advertisements
Advertisements
What is the role of coke in the extraction of iron from its oxides?
Concept: undefined > undefined
Arrange the following in the increasing order of property mentioned :
H3PO3, H3PO4, H3PO2 (Reducing character)
Concept: undefined > undefined
Advertisements
How can you distinguish between propanal and propanone?
Concept: undefined > undefined
Indicate the principle behind the method used for the refining of zinc.
Concept: undefined > undefined
What is the role of silica in the extraction of copper?
Concept: undefined > undefined
Which form of the iron is the purest form of commercial iron?
Concept: undefined > undefined
For the hydrolysis of methyl acetate in aqueous solution, the following results were obtained :
| t/s | 0 | 30 | 60 |
| [CH3COOCH3] / mol L–1 | 0.60 | 0.30 | 0.15 |
(i) Show that it follows pseudo first order reaction, as the concentration of water remains constant.
(ii) Calculate the average rate of reaction between the time interval 30 to 60 seconds.
(Given log 2 = 0.3010, log 4 = 0.6021)
Concept: undefined > undefined
For a chemical reaction R → P, the variation in the concentration (R) vs. time (t) plot is given as:
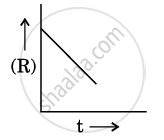
(i) Predict the order of the reaction.
(ii) What is the slope of the curve ?
(iii) Write the unit of rate constant for this reaction.
Concept: undefined > undefined
The following data were obtained during the first order thermal decomposition of SO2Cl2 at a constant volume :
SO2Cl2 (g) → SO2 (g) + Cl2 (g)
| Experiment | Time/s–1 | Total pressure/atm |
| 1 | 0 | 0.4 |
| 2 | 100 | 0.7 |
Calculate the rate constant.
(Given : log 4 = 0.6021, log 2 = 0.3010)
Concept: undefined > undefined
Give reasons for the following : (CH3)3 P = O exists but (CH3)3 N = O does not.
Concept: undefined > undefined
Describe the roles of the following: SiO2 in the extraction of copper from copper matte
Concept: undefined > undefined
Name the method used for refining of copper metal.
Concept: undefined > undefined
Classify the following solids in different categories based on the nature of intermolecular forces operating in them:
Potassium sulphate, tin, benzene, urea, ammonia, water, zinc sulphide, graphite, rubidium, argon, silicon carbide.
Concept: undefined > undefined
Classify each of the following solids as ionic, metallic, molecular, network (covalent) or amorphous.
(i) Tetra phosphorus decoxide (P4O10)
(ii) Ammonium phosphate (NH4)3PO4
(iii) SiC
(iv) I2
(v) P4
(vii) Graphite
(viii) Brass
(ix) Rb
(x) LiBr
(xi) Si
Concept: undefined > undefined
For a reaction, \[\ce{A + B -> Product}\]; the rate law is given by, `r = k[A]^(1/2)[B]^2`. What is the order of the reaction?
Concept: undefined > undefined
The conversion of molecules X to Y follows second order kinetics. If concentration of X is increased to three times how will it affect the rate of formation of Y?
Concept: undefined > undefined
From the rate expression for the following reaction, determine the order of reaction and the dimension of the rate constant.
\[\ce{3NO_{(g)} -> N2O_{(g)}}\] Rate = k[NO]2
Concept: undefined > undefined
From the rate expression for the following reaction, determine the order of reaction and the dimension of the rate constant.
\[\ce{H2O2_{( aq)} + 3I^-_{( aq)} + 2H^+ -> 2H2O_{(l)} + I^-_3}\] Rate = k[H2O2][I−]
Concept: undefined > undefined
From the rate expression for the following reaction, determine the order of reaction and the dimension of the rate constant.
\[\ce{CH3CHO_{(g)} -> CH4_{(g)} + CO_{(g)}}\] Rate = k [CH3CHO]3/2
Concept: undefined > undefined
