Advertisements
Advertisements
प्रश्न
Answer the following question.
a) What is meant by corrosion?
b) Write names of any two methods of prevention of corrosion.
c) In which method, metal like copper, aluminium are coated with a thin layer of their oxides by means of electrolysis.
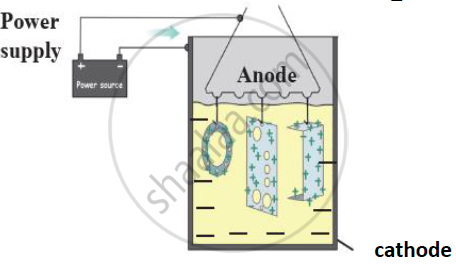
d) Explain this method with diagram.
उत्तर
a. corrosion: Corrosion is a process where the water or the moisture on the surface of the metal oxidizes with the atmospheric oxygen.
b. Methods of prevention (any two each carry 1/2 mark) 1
1. Galvanizing 2. Anodization 3. Tinning
4. Electroplating 5. Alloying
c. Anodization
d. In this process cupper, aluminum are coated with a thin layer of their oxides by means of electrolysis. For this copper or aluminum article is used as anode. It obstructs the contact of the aluminum or copper with oxygen and water.
APPEARS IN
संबंधित प्रश्न
Give two examples of alloys with their chemical composition.
Explain the terms Corrosion
Which metals do not corrode easily?
Two methods by which rusting of iron can be prevented are ______ and ______.
Explain why Iron sheets are coated with zinc during galvanization.
What type of chemical reaction is involved in the corrosion of iron?
Explain why, iron sheets are coated with zinc.
State two conditions for the rusting of iron.
In one method of rust prevention, the iron is not coated with anything. Which is this method?
What is meant by 'rusting of iron'? With the help of labelled diagrams, describe an activity to find out the conditions under which iron rusts.
Explain why, the galvanised iron article is protected against rusting even if the zinc layer is broken.
Brass is an alloy of:
(a) Cu and Sn
(b) Cu and Pb
(c) Pb and Sn
(d) Zn and Cu
A metal X which is resistant to corrosion is produced by the electrolysis of its molten oxide whereas another metal Y which is also resistant to corrosion is produced by the reduction of its oxide with carbon. Metal X can be used in powder form in thermite welding whereas metal Y is used in making cathodes of ordinary dry cells.
(a) Name the metals X and Y.
(b) Which of the two metals is more reactive : X or Y?
(c) Name one ore or metal X. Also write its chemical formula.
(d) Name one ore or metal Y. Also write its chemical formula.
(e) Name one alloy of metal X and one alloy of metal Y.
Bronze is an alloy of ______.
Name the metal which is a constituent of blood pigment?
Name the metal which is a constituent of plant pigment?
Compare roasting and calcination.
Write scientific reasons.
Lemon or tamarind is used for cleaning copper vessels turned greenish.
Identify the process shown in the diagram and explain it in short

Find the odd man out:
Write three methods of preventing rusting of iron.
Give reason.
A wooden article should be polished.
Give reason.
Copper and brass utensils should be tinned.
_______ is an alloy made from iron, carbon and chromium.
Observe the following diagram and give answers.

- Name this method of prevention of corrosion.
- For prevention of which metal this method is used?
- What is used as anode in this method?
What is rust?
State two conditions necessary for rusting of iron.
Amalgam is an alloy of ____________.
Copper objects lose their shine and form green coating of ____________.
Which among the following alloys contain mercury as one of its constituents?
The diagram shows the reaction between metal and dil. acid.

What is the reason for different behaviour of Mg in test tube B?
Marble’s popularity began in ancient Rome and Greece, where white and off-white marble were used to construct a variety of structures, from hand-held sculptures to massive pillars and buildings.

The substance not likely to contain CaCO3 is:
Identify the correct statement from the following:
A man painted his main gate made up of iron, to
- prevent it from rusting.
- protect it from the sun.
- make it look beautiful.
- make it dust-free.
Which of the above statement(s) is/are correct?
The iron pillar near the Qutub Minar in Delhi is famous for the following facts. Which of these facts is responsible for its long stability?
Give scientific reasons.
Silver amalgam is used for filling dental cavities.
