Advertisements
Advertisements
प्रश्न
Find the maximum magnitude of the linear momentum of a photoelectron emitted when a wavelength of 400 nm falls on a metal with work function 2.5 eV.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
उत्तर
Given :-
wavelength of light , `λ = 400 "nm" = 400 xx 10^-9 "m"`
Work function of metal, `phi = 2.5 "eV"`
From Einstein's photoelectric equation,
Kinetic energy = `(hc)/λ - phi`
Here, c = speed of light
h = Planck's constant
`therefore K.E. = (6.63 xx 10^-34 xx 3 xx 10^8)/(4 xx 10^-7 xx 1.6 xx 10^-19) - 2.5 "eV"`
`= 0.605 "eV"`
Also , `K.E. = p^2/(2m)`
where p is momentum and m is the mass of an electron.
`therefore p^2 = 2"m" xx K.E.`
`⇒ p^2 = 2 xx 9.1 xx 10^-31 xx 0.605 xx 1.6 xx 10^-19`
`⇒ p = 4.197 xx 10^-25 "kg - m/s"`
APPEARS IN
संबंधित प्रश्न
Ultraviolet light of wavelength 2271 Å from a 100 W mercury source irradiates a photo-cell made of molybdenum metal. If the stopping potential is −1.3 V, estimate the work function of the metal. How would the photo-cell respond to a high intensity (∼105 W m−2) red light of wavelength 6328 Å produced by a He-Ne laser?
Should the energy of a photon be called its kinetic energy or its internal energy?
It is found that yellow light does not eject photoelectrons from a metal. Is it advisable to try with orange light or with green light?
Two photons of
When the intensity of a light source in increased,
(a) the number of photons emitted by the source in unit time increases
(b) the total energy of the photons emitted per unit time increases
(c) more energetic photons are emitted
(d) faster photons are emitted
A photon of energy hv is absorbed by a free electron of a metal with work-function hv − φ.
An atom absorbs a photon of wavelength 500 nm and emits another photon of wavelength 700 nm. Find the net energy absorbed by the atom in the process.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
A totally reflecting, small plane mirror placed horizontally faces a parallel beam of light, as shown in the figure. The mass of the mirror is 20 g. Assume that there is no absorption in the lens and that 30% of the light emitted by the source goes through the lens. Find the power of the source needed to support the weight of the mirror.
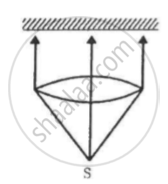
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
A sphere of radius 1.00 cm is placed in the path of a parallel beam of light of large aperture. The intensity of the light is 0.5 W cm−2. If the sphere completely absorbs the radiation falling on it, find the force exerted by the light beam on the sphere.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
Find the maximum kinetic energy of the photoelectrons ejected when light of wavelength 350 nm is incident on a cesium surface. Work function of cesium = 1.9 eV
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
The electric field associated with a monochromatic beam is 1.2 × 1015 times per second. Find the maximum kinetic energy of the photoelectrons when this light falls on a metal surface whose work function is 2.0 eV.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
Answer the following question.
Plot a graph of photocurrent versus anode potential for radiation of frequency ν and intensities I1 and I2 (I1 < I2).
In photoelectric effect, the photoelectric current started to flow. This means that the frequency of incident radiations is ______.
In photoelectric effect the photo current ______.
Do all the electrons that absorb a photon come out as photoelectrons?
The graph shows the variation of photocurrent for a photosensitive metal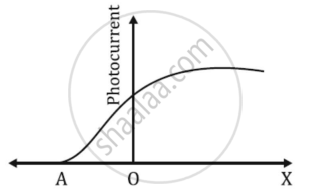
- What does X and A on the horizontal axis represent?
- Draw this graph for three different values of frequencies of incident radiation ʋ1, ʋ2 and ʋ3 (ʋ3 > ʋ2 > ʋ1) for the same intensity.
- Draw this graph for three different values of intensities of incident radiation I1, I2 and I3 (I3 > I2 > I1) having the same frequency.
Why it is the frequency and not the intensity of the light source that determines whether the emission of photoelectrons will occur or not? Explain.
How would the stopping potential for a given photosensitive surface change if the intensity of incident radiation was decreased? Justify your answer.
How would the stopping potential for a given photosensitive surface change if the frequency of the incident radiation were increased? Justify your answer.
A metallic plate exposed to white light emits electrons. For which of the following colours of light, the stopping potential will be maximum?
