Advertisements
Advertisements
प्रश्न
For a chemical reaction R → P, the variation in the concentration (R) vs. time (t) plot is given as:
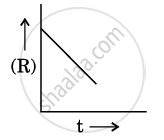
(i) Predict the order of the reaction.
(ii) What is the slope of the curve ?
(iii) Write the unit of rate constant for this reaction.
(iii) Write the unit of rate constant for this reaction.
उत्तर १
For a chemical reaction R→P, the variation in the concentration (R) vs. time (t) plot is given as follows:
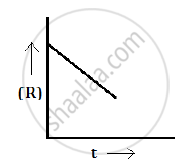
(i) The order of the reaction zero.
(ii) Slope = -k
उत्तर २
(i) The variation in the concentration (R) vs. time (t) plot shown here represents a zero order reaction, for which the rate of the reaction is proportional to zero power of the concentration of the reactants.
(ii) For a zero order reaction, rate constant is given as
`k=([R]_0-[R])/t`
So, the slope of the curve for the variation in the concentration (R) vs. time (t) plot is equal to the negative of the rate constant for the reaction.
उत्तर ३
(iii) Since it is a first order reaction, the unit of the rate constant is s−1.
APPEARS IN
संबंधित प्रश्न
A reaction is second order in A and first order in B.
(i) Write the differential rate equation.
(ii) How is the rate affected on increasing the concentration of A three times?
(iii) How is the rate affected when the concentrations of both A and B are doubled?
For a reaction: 
Rate = k
(i) Write the order and molecularity of this reaction.
(ii) Write the unit of k.
Write two factors that affect the rate of reaction.
For the hydrolysis of methyl acetate in aqueous solution, the following results were obtained :
| t/s | 0 | 30 | 60 |
| [CH3COOCH3] / mol L–1 | 0.60 | 0.30 | 0.15 |
(i) Show that it follows pseudo first order reaction, as the concentration of water remains constant.
(ii) Calculate the average rate of reaction between the time interval 30 to 60 seconds.
(Given log 2 = 0.3010, log 4 = 0.6021)
From the rate expression for the following reaction, determine the order of reaction and the dimension of the rate constant.
\[\ce{CH3CHO_{(g)} -> CH4_{(g)} + CO_{(g)}}\] Rate = k [CH3CHO]3/2
A reaction is first order in A and second order in B. How is the rate affected on increasing the concentration of B three times?
Define the following terms:
Pseudo first-order reaction
In any unimolecular reaction:
(i) only one reacting species is involved in the rate determining step.
(ii) the order and the molecularity of slowest step are equal to one.
(iii) the molecularity of the reaction is one and order is zero.
(iv) both molecularity and order of the reaction are one.
Assertion: Rate constants determined from Arrhenius equation are fairly accurate for simple as well as complex molecules.
Reason: Reactant molecules undergo chemical change irrespective of their orientation during collision.
A flask contains a mixture of compounds A and B. Both compounds decompose by first-order kinetics. The half-lives for A and B are 300 s and 180 s, respectively. If the concentrations of A and B are equal initially, the time required for the concentration of A to be four times that of B (in s) is ______. (Use ln 2 = 0.693)
