Advertisements
Advertisements
प्रश्न
From the rate expression for the following reaction, determine the order of reaction and the dimension of the rate constant.
\[\ce{CH3CHO_{(g)} -> CH4_{(g)} + CO_{(g)}}\] Rate = k [CH3CHO]3/2
उत्तर
Given rate = k [CH3CHO]3/2
Therefore, order of reaction = `3/2`
Dimension of k = `"Rate"/["CH"_3"CHO"]^(3/2)`
= `("mol L"^-1 "s"^(-1))/("mol L"^(-1))^(3/2)`
= `"L"^(1/2) "mol"^(-1/2) "s"^(-1)`
APPEARS IN
संबंधित प्रश्न
For a chemical reaction R → P, the variation in the concentration (R) vs. time (t) plot is given as:
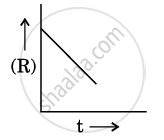
(i) Predict the order of the reaction.
(ii) What is the slope of the curve ?
(iii) Write the unit of rate constant for this reaction.
The following data were obtained during the first order thermal decomposition of SO2Cl2 at a constant volume :
SO2Cl2 (g) → SO2 (g) + Cl2 (g)
| Experiment | Time/s–1 | Total pressure/atm |
| 1 | 0 | 0.4 |
| 2 | 100 | 0.7 |
Calculate the rate constant.
(Given : log 4 = 0.6021, log 2 = 0.3010)
The conversion of molecules X to Y follows second order kinetics. If concentration of X is increased to three times how will it affect the rate of formation of Y?
From the rate expression for the following reaction, determine the order of reaction and the dimension of the rate constant.
\[\ce{H2O2_{( aq)} + 3I^-_{( aq)} + 2H^+ -> 2H2O_{(l)} + I^-_3}\] Rate = k[H2O2][I−]
From the rate expression for the following reaction, determine the order of reaction and the dimension of the rate constant.
\[\ce{C2H5Cl_{(g)} -> C2H4_{(g)} + HCl_{(g)}}\] Rate = k [C2H5Cl]
A reaction is second order with respect to a reactant. How is the rate of reaction affected if the concentration of the reactant is doubled?
A reaction is first order in A and second order in B. Write the differential rate equation.
A reaction is first order in A and second order in B. How is the rate affected on increasing the concentration of B three times?
In a reaction between A and B, the initial rate of reaction (r0) was measured for different initial concentrations of A and B as given below:
| A/mol L−1 | 0.20 | 0.20 | 0.40 |
| B/mol L−1 | 0.30 | 0.10 | 0.05 |
| r0/mol L−1 s−1 | 5.07 × 10−5 | 5.07 × 10−5 | 1.43 × 10−4 |
What is the order of the reaction with respect to A and B?
Which of the following statements is not correct about order of a reaction.
The value of rate constant of a pseudo first order reaction ______.
Why does the rate of any reaction generally decreases during the course of the reaction?
Assertion: Order and molecularity are same.
Reason: Order is determined experimentally and molecularity is the sum of the stoichiometric coefficient of rate determining elementary step.
Assertion: The enthalpy of reaction remains constant in the presence of a catalyst.
Reason: A catalyst participating in the reaction, forms different activated complex and lowers down the activation energy but the difference in energy of reactant and product remains the same.
If the 0.05 molar solution of m+ is replaced by a 0.0025 molar m+ solution, then the magnitude of the cell potential would be
For a reaction 1/2 A ⇒ 2B, rate of disappearance of A is related 't o the appearance of B by the expression:
For reaction 2A + B → BC + D which of the following does not Express the reaction rates
At concentration of 0.1 and 0.2 mol L–1 the rates of deem position of a compound were found to be 0.18 and 0.72 mol L–1 m–1. What is the order of the reaction?
Assertion (A): Order of reaction is applicable to elementary as well as complex reactions.
Reason (R): For a complex reaction, molecularity has no meaning.
Which of the following statement is true?
