Advertisements
Advertisements
प्रश्न
Why cannot aniline be prepared by Gabriel phthalimide synthesis?
उत्तर
- Gabriel phthalimide synthesis is used for the preparation of aliphatic primary amines.
- It involves the nucleophilic substitution of alkyl halides by the anion formed by the phthalimide.
- But aryl halides do not undergo nucleophilic substitution with the anion formed by the phthalimide.
Hence, aniline cannot be prepared by Gabriel phthalimide synthesis.
APPEARS IN
संबंधित प्रश्न
How is ethyl amine prepared from methyl iodide?
How are propan-1-amine and propan-2-amine prepared from oxime?
Illustrate the following reaction giving suitable example in each case:Gabriel phthalimide synthesis
How do you convert the following: C6H5CONH2 to C6H5NH2
Give the structures of A, B and C in the following reactions :
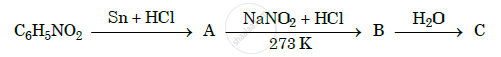
Give the structure of A, B and C in the following reaction:
\[\ce{C6H5N2Cl ->[CuCN] A ->[H2O/H+] B ->[NH3][\Delta] C}\]
Give the structures of A, B and C in the following reaction:
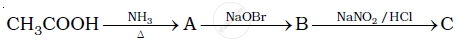
Give the structures of A, B and C in the following reaction.

Explain the mechanism of action of hydroiodic acid on 3-methylbutan-2-ol.
Arrange the following in the increasing order of their pKb values:
C6H5NH2, C2H5NH2, C6H5NHCH3
Give the structures of A, B and C in the following reactions :

Write structures of compounds A and B in each of the following reactions: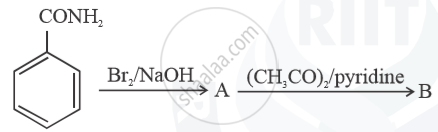
Answer the following
Identify A and B in the following reactions.
\[\ce{C6H5CH2Br->[alco.][KCN]A ->[Na/ethanol]B.}\]
Answer the following
Explain the ammonolysis of alkyl halides.
Write reactions to prepare ethanamine from Acetonitrile.
The following amines is the product of Gabriel phthalimide synthesis.
Write the order of reactivity of alkyl halides with ammonia.
Write reactions to bring about the following conversions.
Acetamide to methylamine
The end product C of the following reaction is
\[\ce{C2H5NH2 ->[HNO2] A ->[PCl5] B ->[NH3][Alcohol] C}\]
Identify the product obtained when benzyl chloride undergoes ammonolysis in presence of excess ammonia followed by the reaction with two moles of methyl iodide.
What is the molar mass of the amine formed when acetamide undergoes Hofmann bromamide degradation?
Which nitrogen containing compound amongst the following would undergo Mendius reduction to furnish primary amine \[\ce{(R - NH2)}\]?
The reduction of alkyl cyanide with sodium and ethanol to give primary amines is, ____________.
Identify the INCORRECT statement regarding Hofmann bromamide reaction.
In aqueous phase the order of basic strength of alkylamine is ______.
Which of the following reagents is used in Mendius reduction reaction of alkyl cyanide?
Identify product B in the following reaction.
\[\ce{Aniline ->[NaNO2][HCl] A ->[KI] B}\]
Which of the following does NOT give carbylamine test?
In order to prepare a 1° amine from an alkyl halide with simultaneous addition of one \[\ce{CH2}\] group in the carbon chain, the reagent used as source of nitrogen is ______.
The source of nitrogen in Gabriel synthesis of amines is ______.
Among the following amines, the strongest Brönsted base is:
Which of the following methods of preparation of amines will give same number of carbon atoms in the chain of amines as in the reactant?
How will you carry out the following conversion?
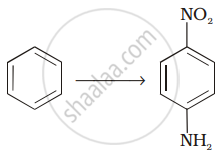
How will you carry out the following conversion?
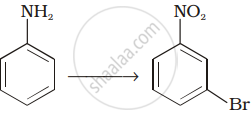
How will you carry out the following conversions?
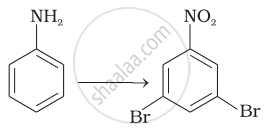
Describe Gabriel's phthalimide synthesis. (Give reaction)
A primary amine is formed by an amide on treatment with bromine and alkali. The primary amine has
Ethylamine can be prepared by the action of bromine and caustic potash on which compound?
Reduction of nitro alkanes yields which compound?
Acetamide and ethyl amide can be distinguished by reacting with.
C6H5CONHCH3 can be converted into C6H5CH2NHCH3 by:-
Which of the following CANNOT be prepared by ammonolysis of alkyl halide?
A compound 'A' on reduction with iron scrap and hydrochloric acid gives compound 'B' with molecular formula C6H7N. Compound 'B' on reaction with CHCl3 and alcoholic KOH produces an obnoxious smell of carbylamine due to the formation of 'C'. Identify 'A', 'B' and 'C' and write the chemical reactions involved.
- Phenyl methenamine
- N, N - Dimethylaniline
- N - Methyl aniline
- Benzenamine
Choose the correct order of the basic nature of the above amines.
The amine 'A' when treated with nitrous acid gives yellow oily substance. The amine A is ______.
Amides can be converted into amines by the reaction named ______.
Identify the compo ds A and B in the following reactions:
\[\ce{A ->[Nitrating mixture] B ->[(i) Sn/cone. HCI][(ii) NaOH] Aniline}\]
Identify A and B in the following reaction.
\[\ce{C6H5CH2Br ->[Alco.][KCN] A ->[Na/Ethanol][reduction] B}\]
Write a short note on Ammonolysis.
Write a short note on Ammonolysis.
