Advertisements
Advertisements
प्रश्न
Write the structure of the major organic product in the following reaction:
\[\ce{CH3CH2Br + KCN ->[aq.ethanol]}\]
उत्तर
\[\ce{\underset{Bromoethane}{CH3CH2Br} + KCN ->[aq.ethanol][(nucleophilic substitution)] \underset{Propanenitrile}{CH3CH2CN} + KBr}\]
APPEARS IN
संबंधित प्रश्न
Discuss the mechanism of alkaline hydrolysis of bromomethane.
In the following pair of halogen compounds, which compound undergoes a faster SN1 reaction?
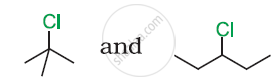
What are ambident nucleophiles? Explain with an example.
What happens when methyl chloride is treated with KCN?
How the following conversion can be carried out?
Ethyl chloride to propanoic acid
AgCN reacts with haloalkanes to form isocyanide. Haloalkanes react with KCN to form alkyl cyanides as the main product. Why?
In the reaction, \[\ce{R - X + NaOR' -> ROR’ + X}\] ( – ve ion). The main product formed is:
Which of the following is a primary halide?
SN1 reaction of alkyl halides lead to ___________.
The increasing order of reactivity towards SN1 mechanism is:
(I) \[\begin{array}{cc}
\ce{CH3-CH-CH2-CH3}\\
|\phantom{........}\\
\ce{CH3}\phantom{.....}
\end{array}\]
(II) CH3CH2CH2Cl
(III) P–CH3O–C6H4–CH2Cl
Complete the following analogy:
Same molecular formula but different structures: A : : Non superimposable mirror images: B
Which of the following statements are correct about the kinetics of this reaction?
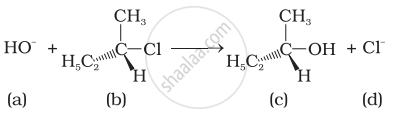
(i) The rate of reaction depends on the concentration of only (b).
(ii) The rate of reaction depends on concentration of both (a) and (b).
(iii) Molecularity of reaction is one.
(iv) Molecularity of reaction is two.
Why are aryl halides less reactive towards nucleophilic substitution reactions than alkyl halides? How can we enhance the reactivity of aryl halides?
When CH3CH2CHCl2 is treated NaNH2 product formed is:-
The major product formed in the following reaction is:
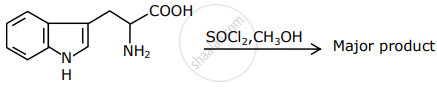
Racemisation occurs in ______.
Arrange the following compounds in increasing order of reactivity towards SN2 reaction.
2-Bromopentane, 1-Bromopentane, 2-Bromo-2-methylbutane
Give the mechanism of the following reaction:
\[\ce{CH3CH2OH ->[H2SO4][413 K] CH3CH2-O-CH2CH3 + H2O}\]
Discuss the mechanism of alkaline hydrolysis of methyl bromide.
