Advertisements
Advertisements
प्रश्न
From the rate expression for the following reaction, determine the order of reaction and the dimension of the rate constant.
\[\ce{H2O2_{( aq)} + 3I^-_{( aq)} + 2H^+ -> 2H2O_{(l)} + I^-_3}\] Rate = k[H2O2][I−]
उत्तर
Given rate = k[H2O2][I−]
Therefore, order of the reaction = 2
Dimension of k = `"Rate"/(["H"_2"O"_2]["I"^(-)])`
= `("mol L"^(-1) "s"^(-1))/(("mol L"^(-1))("mol L"^(-1)))`
= L mol−1 s−1
APPEARS IN
संबंधित प्रश्न
In a first order reaction x → y, 40% of the given sample of compound remains unreacted in 45 minutes. Calculate rate constant of the reaction.
For the first order thermal decomposition reaction, the following data were obtained:
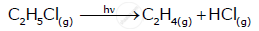
Time / sec Totalpressure / atm
0 0.30
300 0.50
Calculate the rate constant
(Given: log 2 = 0.301, log3 = 0.4771, log 4 = 0.6021)
For the reaction: \[\ce{2A + B → A2B}\] the rate = k[A][B]2 with k = 2.0 × 10−6 mol−2 L2 s−1. Calculate the initial rate of the reaction when [A] = 0.1 mol L−1, [B] = 0.2 mol L−1. Calculate the rate of reaction after [A] is reduced to 0.06 mol L−1.
A reaction is first order in A and second order in B. Write the differential rate equation.
Write resonating structures of ozone.
Write the principle behind the following methods of refining:
Hydraulic washing
Define the following terms:
Half-life period of reaction (t1/2).
Rate law for the reaction \[\ce{A + 2B -> C}\] is found to be Rate = k [A][B]. Concentration of reactant ‘B’ is doubled, keeping the concentration of ‘A’ constant, the value of rate constant will be ______.
For a complex reaction:
(i) order of overall reaction is same as molecularity of the slowest step.
(ii) order of overall reaction is less than the molecularity of the slowest step.
(iii) order of overall reaction is greater than molecularity of the slowest step.
(iv) molecularity of the slowest step is never zero or non interger.
Why can we not determine the order of a reaction by taking into consideration the balanced chemical equation?
Match the graph given in Column I with the order of reaction given in Column II. More than one item in Column I may link to the same item of Column II.
| Column I | Column II | |
| (i) |  |
|
| (ii) |  |
(a) 1st order |
| (iii) |  |
(b) Zero-order |
| (iv) |  |
A catalyst in a reaction changes which of the following?
For a reaction A + B → products, the rate law is given by: r = `K[A]^(1/2)`. What is the order of reaction?
For a reaction R → p the concentration of reactant change from 0.03 m to 0.02 m in minute, calculate the average rate of the reaction using the unit of second.
For a reaction \[\ce{Cl2l(g) + 2No(g) -> 2NaCl(g)}\] the rate law is expressed as rate= K[Cl2] [No]2 what is the order of the reaction?
For reaction 2A + B → BC + D which of the following does not Express the reaction rates
The half-life period of a. substance in a certain enzyme catalysed reaction is 138 s. The time required for the concentration of the substance to fall from 1.28 mol–1 to 0.04 mg L–1 is
Identify the order of reaction from the following unit for its rate constant:
L mol–1s–1
