Advertisements
Advertisements
प्रश्न
How is Benzonitrile converted to Benzophenone?
How is benzophenone prepared from benzonitrile?
उत्तर
Benzonitrile reacts with phenyl magnesium bromide in equimolecular proportion in the presence of dry ether to give an adduct, which on acid hydrolysis gives benzophenone

APPEARS IN
संबंधित प्रश्न
Classify the following ligands into monodentate and polydentate —
- Ammonia
- Carbon monoxide
- Ethylene diamine
- Ethylene diamine tetra acetate ion
Explain the following, giving two examples:
Coordination entity
Give the oxidation state, d-orbital occupation and coordination number of the central metal ion in the following complex:
K3[Co(C2O4)3]
Which of the following compounds is used as a semipermeable membrane?
(a) Potassium ferrocyanide
(b) Potassium argentocyanide
(c) Sodium meta aluminate
(d) Copper ferrocyanide
What happens when PCl5 is heated? Write the equations involved.
Complete the following reactions
NH3+3Cl2(excess) ---->
Write the IUPAC names of the following coordination compounds:
[Cr(NH3)4(H2O)2]Cl3
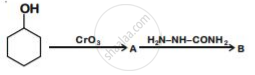
Write structures of compounds A and B of the following reaction :

Write structures of compounds A and B of the following reaction :
Which of the following represents a chelate ligand?
Ligand (en) is an example of ___________.
A group of atoms can function as a ligand only when:
The oxidation number of Fe in K4[Fe(CN)6] is ____________.
In which of the following compounds, the Central metal atom/ion is in the lowest oxidation state?
When 1 mol \[\ce{CrCl3.6H2O}\] is treated with excess of \[\ce{AgNO3}\], 3 mol of \[\ce{AgCl}\] are obtained. The formula of the complex is ______.
Identify the correct statements for the behaviour of ethane-1, 2-diamine as a ligand.
(i) It is a neutral ligand.
(ii) It is a didentate ligand.
(iii) It is a chelating ligand.
(iv) It is a unidentate ligand.
Assertion: \[\ce{Cr(H2O)6]Cl2 and [Fe(H2O)6]Cl2}\] are reducing in nature.
Reason: Unpaired electrons are present in their d-orbitals.
What is the relationship between observed colour of the complex and the wavelength of light absorbed by the complex?
Which of the following ligands can exhibit linkage isomerism?
Metal attached with EDTA in an octahedral complex, has coordination number ______.
Why chelate complexes are more stable than complexes with unidentate ligands?
How many ions are produced from the complex Co(NH3)6]Cl2 in solution?
Given below are two statements.
- Statement I: In CuSO4 · 5H2O, Cu-O bonds are present.
- Statement II: In CuSO4 · 5H2O, ligands coordinating with Cu(II) ion are O- and S based ligands.
In light of the above statement, choose the correct answer from the options given below.
The correct order of the spin-only magnetic moment of metal ions in the following low spin complexes,
\[\ce{[V(CN)6]^{4-}, [Fe(CN)6]^{4-}, [Ru(NH3)6]^{3+} and [Cr(NH3)6]^2}\] is:
The one that will show optical activity is: (en = ethane 1, 2-diamine)
Does ionization isomer for the following compound exist? Justify your answer.
\[\ce{Hg[Co(SCN)4]}\]
Explain the following, giving two examples:
Coordination number
Give two examples of unidentate ligand.
