Advertisements
Advertisements
Question
A charged capacitor of capacitance C is discharged through a resistance R. A radioactive sample decays with an average-life τ. Find the value of R for which the ratio of the electrostatic field energy stored in the capacitor to the activity of the radioactive sample remains constant in time.
Solution
Discharging of a capacitor through a resistance R is given by
`Q = qe^-t"/"CR`
Here, Q = Charge left
q = Initial charge
C = Capacitance
R = Resistance
Energy , `E = 1/2 Q^2/C = (q^2e^(-2t"/"CR))/(2C)`
Activity , `A = A_0e^(-lambdat)`
Here , `A_0` = Initial activity
`lambda` = Disintegration constant
∴ Ratio of the energy to the activity = `E/A = (q^2 xx e^(-2t"/"CR))/(2CA_0e^(-lambdat)`
Since the terms are independent of time, their coefficients can be equated.
`(2t)/(CR) = lambdat`
⇒ `lambda = 2/(CR)`
⇒`1/tau = 2/(CR)`
⇒ `R = 2 tau/C`
APPEARS IN
RELATED QUESTIONS
The half life of a certain radioactive material against \u0003α-decay is 100 days. After how much time, will the undecayed fraction of the material be 6.25%?
Write nuclear reaction equation for α-decay of `""_94^242"Pu"`.
Write nuclear reaction equation for β−-decay of `""_15^32"P"`.
Write nuclear reaction equation for β−-decay of `""_83^210"Bi"`.
Write nuclear reaction equation for β+-decay of `""_6^11"C"`.
Define ‘activity’ of a radioactive material and write its S.I. units.
Plot a graph showing variation of activity of a given radioactive sample with time.
The sequence of stepwise decay of a radioactive nucleus is
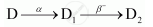
If the atomic number and mass number of D2 are 71 and 176 respectively, what are their corresponding values of D?
State the law of radioactive decay. hence derive the relation N = Noe-λt . Represent it graphically.
The half-life of 199Au is 2.7 days. (a) Find the activity of a sample containing 1.00 µg of 198Au. (b) What will be the activity after 7 days? Take the atomic weight of 198Au to be 198 g mol−1.
A certain sample of a radioactive material decays at the rate of 500 per second at a certain time. The count rate falls to 200 per second after 50 minutes. (a) What is the decay constant of the sample? (b) What is its half-life?
The half-life of 226Ra is 1602 y. Calculate the activity of 0.1 g of RaCl2 in which all the radium is in the form of 226Ra. Taken atomic weight of Ra to be 226 g mol−1 and that of Cl to be 35.5 g mol−1.
The half-life of a radioisotope is 10 h. Find the total number of disintegration in the tenth hour measured from a time when the activity was 1 Ci.
A vessel of volume 125 cm3 contains tritium (3H, t1/2 = 12.3 y) at 500 kPa and 300 K. Calculate the activity of the gas.
4 × 1023 tritium atoms are contained in a vessel. The half-life of decay tritium nuclei is 12.3 y. Find (a) the activity of the sample, (b) the number of decay in the next 10 hours (c) the number of decays in the next 6.15 y.
`""_83^212"Bi"` can disintegrate either by emitting an α-particle of by emitting a β−-particle. (a) Write the two equations showing the products of the decays. (b) The probabilities of disintegration α-and β-decays are in the ratio 7/13. The overall half-life of 212Bi is one hour. If 1 g of pure 212Bi is taken at 12.00 noon, what will be the composition of this sample at 1 P.m. the same day?
Plot a graph showing the variation of undecayed nuclei N versus time t. From the graph, find out how one can determine the half-life and average life of the radioactive nuclei.
Half life of a certain radioactive material is 8 hours.
If one starts with 600 g of this substance, how much of it will disintegrate in one day?
A nucleus with Z = 92 emits the following in a sequence:
α, β‾, β‾, α, α, α, α, α, β‾, β‾, α, β+, β+, α
Then Z of the resulting nucleus is ______.
