Advertisements
Advertisements
Question
Find out the type of isomerism exhibited by the following pair.
CH3 – CH2 – NH – CH2 - CH3 and CH3 - NH - CH2 - CH2 - CH3
Solution
Metamerism
APPEARS IN
RELATED QUESTIONS
Write IUPAC name of the product obtained by the ozonolysis of the following compound:
3,4-Dimethyl-hept-3-ene
Draw the cis and trans structures of hex-2-ene. Which isomer will have higher b.p. and why?
What effect does branching of an alkane chain has on its boiling point?
What is the relationship between the members of following pairs of structures? Are they structural or geometrical isomers or resonance contributors?
\[\begin{array}{cc}
\ce{D}\phantom{......}\ce{H}\\
\backslash\phantom{......}/\\
\ce{C = C}\\
\phantom{...}/\phantom{......}\backslash\phantom{...}\\\ce{H}\phantom{.......}\ce{D}
\end{array}\]
\[\begin{array}{cc}
\ce{D}\phantom{......}\ce{D}\\
\backslash\phantom{......}/\\
\ce{C = C}\\
\phantom{...}/\phantom{......}\backslash\phantom{...}\\\ce{H}\phantom{.......}\ce{H}\end{array}\]
Find out the type of isomerism exhibited by the following pair.
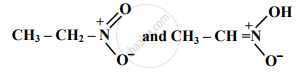
Find out the type of isomerism exhibited by the following pair.

What type(s) of isomerism is(are) shown by [Co(NH3)4Br2]Cl?
But-1-ene and But-2-ene are examples of ____________.
Which of the following is a functional isomer of pentan-2-ol?
What is the relationship between the members of following pairs of structures? Are they structural or geometrical isomers or resonance contributors?

What is the relationship between the members of following pairs of structures? Are they structural or geometrical isomers or resonance contributors?
\[\begin{array}{cc}\ce{^+OH}\\||\\
\ce{H - C - OH}\end{array}\]
\[\begin{array}{cc}\ce{OH}\phantom{.}\\|\phantom{...}\\
\ce{H - C^+ - OH}\end{array}\]
Which of the following pairs are position isomers?
| I. | \[\begin{array}{cc} \phantom{.......................}\ce{O}\\ \phantom{.......................}||\\ \ce{CH3 - CH2 - CH2 - CH2 - C - H} \end{array}\] |
| II. | \[\begin{array}{cc} \phantom{.................}\ce{O}\\ \phantom{.................}||\\ \ce{CH3 - CH2 - CH2 - C - H} \end{array}\] |
| III. | \[\begin{array}{cc} \ce{CH3 - CH2 - C - CH2 - CH3}\\ \phantom{}||\\ \phantom{}\ce{O} \end{array}\] |
| IV. | \[\begin{array}{cc} \ce{CH3 - CH - CH2 - C - H}\\ \phantom{...}|\phantom{............}||\phantom{}\\ \phantom{...}\ce{CH3}\phantom{.........}\ce{O}\phantom{} \end{array}\] |
(i) I and II
(ii) II and III
(iii) II and IV
(iv) III and IV
Which of the following pairs are not functional group isomers?
| I. | \[\begin{array}{cc} \phantom{.......................}\ce{O}\\ \phantom{.......................}||\\ \ce{CH3 - CH2 - CH2 - CH2 - C - H} \end{array}\] |
| II. | \[\begin{array}{cc} \phantom{.................}\ce{O}\\ \phantom{.................}||\\ \ce{CH3 - CH2 - CH2 - C - H} \end{array}\] |
| III. | \[\begin{array}{cc} \ce{CH3 - CH2 - C - CH2 - CH3}\\ \phantom{}||\\ \phantom{}\ce{O} \end{array}\] |
| IV. | \[\begin{array}{cc} \ce{CH3 - CH - CH2 - C - H}\\ \phantom{...}|\phantom{............}||\phantom{}\\ \phantom{...}\ce{CH3}\phantom{.........}\ce{O}\phantom{} \end{array}\] |
(i) II and III
(ii) II and IV
(iii) I and IV
(iv) I and II
Consider structures I to VII and answer the question:
| I. | CH3 – CH2 – CH2 – CH2 – OH |
| II. | \[\begin{array}{cc} \ce{CH3 - CH2 - CH - CH3}\\ \phantom{.....}|\\ \phantom{.......}\ce{OH} \end{array}\] |
| III. | \[\begin{array}{cc} \phantom{...}\ce{CH3}\\ \phantom{}|\\ \ce{CH3 - C - CH3}\\ \phantom{}|\\ \phantom{..}\ce{OH} \end{array}\] |
| IV. | \[\begin{array}{cc} \ce{CH3 - CH - CH2 - OH}\\ |\phantom{........}\\ \ce{CH3}\phantom{......} \end{array}\] |
| V. | CH3 – CH2 – O – CH2 – CH3 |
| VI. | CH3 – O – CH2 – CH2 – CH3 |
| VII. | \[\begin{array}{cc} \ce{CH3 - O - CH - CH3}\\ \phantom{...}|\\ \phantom{......}\ce{CH3} \end{array}\] |
Identify the pairs of compounds which are functional group isomers.
Compounds with same molecular formula but differing in their structures are said to be structural isomers. What type of structural isomerism is shown by
CH3 – S – CH2 – CH2 – CH3
And
\[\begin{array}{cc}
\phantom{.....................}\ce{CH3}\\
\phantom{................}/\\
\phantom{}\ce{CH3 - S - CH}\\
\phantom{...............}\backslash\\
\phantom{....................}\ce{CH3}
\end{array}\]
Assertion (A): Pent- 1- ene and pent- 2- ene are position isomers.
Reason (R): Position isomers differ in the position of functional group or a substituent.
The compound which shows metamerism is ______
The correct stereochemical name of
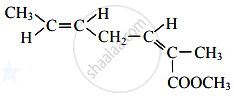
How many structural isomers possible of the molecular formula C3H6O (excluding enol form)?
Compound with molecular formula C3H6O can show ______.
Which of the following reactions will not produce a racemic product?
The total number of possible isomers of the complex compound [CuII(NH3)4][PtIICl4] is ______.
Which of the following pairs of compounds is an example of position isomerism?
