Advertisements
Advertisements
Question
Identify the order of reaction from the following unit for its rate constant:
L mol–1s–1
Solution
The unit L mol–1sec–1 for rate constant is the unit of second-order reaction.
APPEARS IN
RELATED QUESTIONS
Write molecularity of the following reaction:
2NO(g)+O2(g)→2NO2(g)
Write two factors that affect the rate of reaction.
For a reaction : 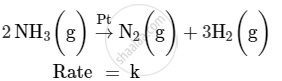
(i) Write the order and molecularity of this reaction.
(ii) Write the unit of k.
A reaction is first order in A and second order in B. Write the differential rate equation.
How does calcination differ from roasting?
Write resonating structures of ozone.
Assertion: Order of the reaction can be zero or fractional.
Reason: We cannot determine order from balanced chemical equation.
Assertion: Order and molecularity are same.
Reason: Order is determined experimentally and molecularity is the sum of the stoichiometric coefficient of rate determining elementary step.
A catalyst in a reaction changes which of the following?
For a reaction \[\ce{Cl2l(g) + 2No(g) -> 2NaCl(g)}\] the rate law is expressed as rate= K[Cl2] [No]2 what is the order of the reaction?
