Advertisements
Advertisements
Question
Number of faradays of electricity required to liberate 12 g of hydrogen is:
Options
1
8
12
16
Solution
12
- The molecular mass of Hydrogen is 1 gram and has a single electron in its configuration as it is the first most element of the periodic table.
- The 12 grams of Hydrogen contains 12 moles of the electrons in it. Usually, the electricity will be passed through the free electrons.
- The 12 grams of Hydrogen contains 12 electrons which are free electrons.
- Each free electron is capable of conducting 1 Faraday of electricity.
- Since 12 grams of Hydrogen are having 12 electrons in it, we require 12 Faraday of electricity.
APPEARS IN
RELATED QUESTIONS
The charge of how many coulomb is required to deposit 1.0 g of sodium metal (molar mass 23.0 g mol-1) from sodium ions is -
- 2098
- 96500
- 193000
- 4196
96500 coulombs correspond to the charge on how many electrons?
Suggest a list of metals that are extracted electrolytically.
Consider the reaction: \[\ce{Cr2O^{2-}_7 + 14H^+ + 6e^- -> 2Cr^{3+} + 7H2O}\]
What is the quantity of electricity in coulombs needed to reduce 1 mol of \[\ce{Cr2O^{2-}_7}\]?
How much charge is required for the following reduction:
1 mol of \[\ce{Al^{3+}}\] to \[\ce{Al}\]?
How much charge is required for the following reduction:
1 mol of \[\ce{Cu^{2+}}\] to \[\ce{Cu}\]?
Three electrolytic cells A, B, C containing solutions of \[\ce{ZnSO4}\], \[\ce{AgNO3}\] and \[\ce{CuSO4}\], respectively, are connected in series. A steady current of 1.5 amperes was passed through them until 1.45 g of silver deposited at the cathode of cell B. How long did the current flow? What mass of copper and zinc were deposited?
Write any two uses of H2SO4
Explain Faraday’s second law of electrolysis
Calculate the mass of Ag deposited at cathode when a current of 2 amperes was passed through a solution of AgNO3 for 15 minutes.
(Given : Molar mass of Ag = 108 g mol−1 lF = 96500 C mol−1)
Following reactions occur at cathode during the electrolysis of aqueous copper(II) chloride solution :
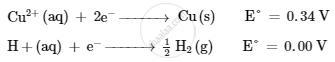
On the basis of their standard reduction electrode potential (E°) values, which reaction is feasible at the cathode and why ?
How much quantity of electricity in coulomb is required to deposit 1.346 × 10-3 kg of Ag in 3.5 minutes from AgNO3 solution?
( Given: Molar mass of Ag is 108 × 10-3 kg mol-1 )
In the electrolysis of aqueous sodium chloride solution which of the half cell reaction will occur at anode?
Aqueous copper sulphate solution and aqueous silver nitrate solution are electrolysed by 1 ampere current for 10 minutes in separate electrolytic cells. Will the mass of copper and silver deposited on the cathode be same or different? Explain your answer.
Time Required to deposite one millimole of aluminium metal by the passage of 9.65 ampere through aqueous solution of aluminium is
When during electrolysis of a solution of Ag No3, 9650 coulombs of charge pass through the electroplating bath, the mass of silver deposite on the cathode will be:-
Given `1/a` = 0.5 CM–1, R = 50 ohm, N = 1.0 then equivalent conductance of electrolytic cell is
On Electrolysis of dilute sulphuric acid using platinum electrodes, the product obtained at the anode will be.
Through an aqueous solution of an unknown salt of metal M (M = 200 g/mol) a current of 1.93 A is passed for 50 min. If 4 g of metal is produced at cathode. The charge on metal ion in solution is ______.
On passing electricity through nitrobenzene solution, it is converted into azobenzene. The mass of azobenzene is ______ mg, if the same quantity of electricity produces oxygen just sufficient to burn 96 mg of fullerene (C60)·
How much electricity in terms of Faraday is required to produce 40.0 g of \[\ce{Al}\] from molten \[\ce{Al2O3}\]?
(Given: Molar mass of Aluminium is 27 g mol−1.)
