Advertisements
Advertisements
Question
The activity R of an unknown radioactive nuclide is measured at hourly intervals. The results found are tabulated as follows:
| t (h) | 0 | 1 | 2 | 3 | 4 |
| R (MBq) | 100 | 35.36 | 12.51 | 4.42 | 1.56 |
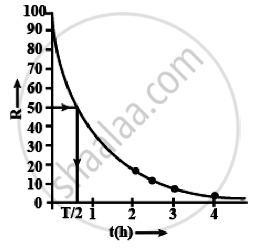
- Plot the graph of R versus t and calculate the half-life from the graph.
- Plot the graph of ln `(R/R_0)` versus t and obtain the value of half-life from the graph.
Solution
We have listed R(MBq) and In `(R/R_0)` in the table below.
| t (h) | 0 | 1 | 2 | 3 | 4 |
| R (MBq) | 100 | 35.36 | 12.51 | 4.42 | 1.56 |
| `R/R_0` | – | – 1.04 | – 2.08 | – 3.11 | – 4.16 |
i. Graph between R versus t is an exponential curve. From the graph at slightly more than `t = 1/2 h` the R should be 50% so at R = 50% the t(h) = 0.7h
= 0.7 × 60 min
= 42 min
ii. The adjacent figure shows the graph of In `(R/R_0)` versus t.
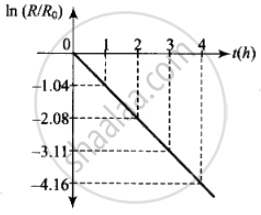
The slope of this graph = – λ
From the graph,
`λ = - ((-4.16 - 3.11)/1) = 1.05 h^-1`
Hence half-life `T_(1/2) = 0.693/λ = 0.693/1.05` = 0.66 h
= 39.6 min ≈ 40 min
APPEARS IN
RELATED QUESTIONS
The half-life of `""_38^90 "Sr"` is 28 years. What is the disintegration rate of 15 mg of this isotope?
Why is it experimentally found difficult to detect neutrinos in this process ?
The masses of 11C and 11B are respectively 11.0114 u and 11.0093 u. Find the maximum energy a positron can have in the β*-decay of 11C to 11B.
(Use Mass of proton mp = 1.007276 u, Mass of `""_1^1"H"` atom = 1.007825 u, Mass of neutron mn = 1.008665 u, Mass of electron = 0.0005486 u ≈ 511 keV/c2,1 u = 931 MeV/c2.)
The decay constant of `""_80^197`Hg (electron capture to `""_79^197`Au) is 1.8 × 10−4 S−1. (a) What is the half-life? (b) What is the average-life? (c) How much time will it take to convert 25% of this isotope of mercury into gold?
The decay constant of 238U is 4.9 × 10−18 S−1. (a) What is the average-life of 238U? (b) What is the half-life of 238U? (c) By what factor does the activity of a 238U sample decrease in 9 × 109 years?
Consider the situation of the previous problem. Suppose the production of the radioactive isotope starts at t = 0. Find the number of active nuclei at time t.
Identify the nature of the radioactive radiations emitted in each step of the decay process given below.
`""_Z^A X -> _Z^A _-1^-4 Y ->_Z^A _-1^-4 W`
Disintegration rate of a sample is 1010 per hour at 20 hours from the start. It reduces to 6.3 x 109 per hour after 30 hours. Calculate its half-life and the initial number of radioactive atoms in the sample.
Which one of the following nuclei has shorter meant life?
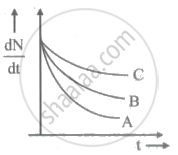
Which sample, A or B shown in figure has shorter mean-life?