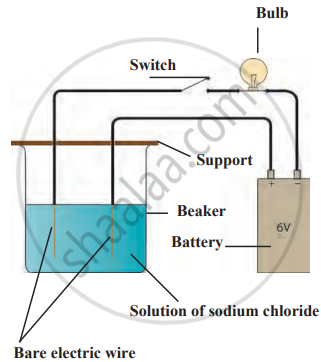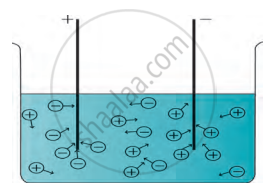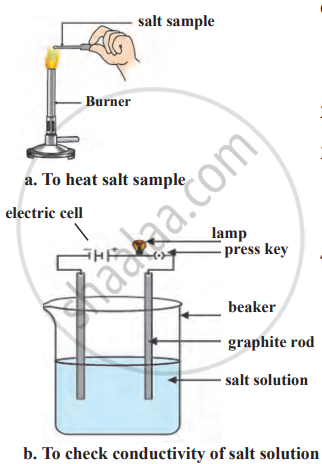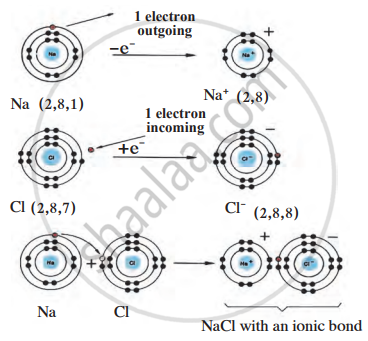
Formation of the compound NaCl Electronic configuration
Atoms are naturally electrically neutral because they have an equal number of protons (positive charge) and electrons (negative charge). However, atoms tend to form ions to achieve a stable electronic configuration, which usually means having a complete octet (eight electrons in the outermost shell).
- Sodium (Na) has one electron in its outermost shell, which makes it unstable. To become stable, it loses this electron, forming a positive ion (Na⁺). This leaves it with a complete octet in the next inner shell.
- Chlorine (Cl) has seven electrons in its outermost shell, which is one short of a complete octet. To become stable, it gains one electron, forming a negative ion (Cl⁻).
Formation of Ionic Bond in Sodium Chloride (NaCl):
When sodium loses one electron, it becomes a cation (Na⁺). When chlorine gains that electron, it becomes an anion (Cl⁻). These oppositely charged ions are then strongly attracted to each other, forming an ionic bond. The result is the stable compound sodium chloride (NaCl), or common table salt.
The stability of ionic compounds like NaCl comes from two factors:
- Complete Octet: Both Na⁺ and Cl⁻ have full outermost shells after the electron transfer, making them stable.
- Strong Ionic Bond: The force of attraction between the positively charged Na⁺ and negatively charged Cl⁻ ions creates a very strong bond, giving the compound high stability.