Advertisements
Advertisements
प्रश्न
Compare the rates of emission of heat by a blackbody maintained at 727°C and at 227°C, if the black bodies are surrounded by an enclosure (black) at 27°C. What would be the ratio of their rates of loss of heat?
उत्तर
Data: T1 = 273 + 727 = 1000 K,
T2 = 273 + 227 = 500 K,
T0 = 273 + 27 = 300 K
(i) The rate of emission of heat, `"dQ"/"dt" = sigma"AT"^4`.
We assume that the surface area A is the same for the two bodies.
`∴ ("dQ"//"dt")_1/("dQ"//"dt")_2 = "T"_1^4/"T"_2^4 = ("T"_1/"T"_2)^4`
`= (1000/500)^4 = 2^4 = 16`
(ii) The rate of loss of heat, `"dQ'"/"dt" = sigma"A" ("T"^4 - "T"_0^4)`
`∴ ("dQ'"//"dt")_1/("dQ'"//"dt")_2 = ("T"_1^4 - "T"_0^4)/("T"_2^4 - "T"_0^4)`
`= (10^12 - 81 xx 10^8)/(625 xx 10^8 - 81 xx 10^8)`
`= ((10000 - 81) xx 10^8)/(544 xx 10^8)`
`= 9919/544 = 18.23`
APPEARS IN
संबंधित प्रश्न
Do you expect the gas in a cooking gas cylinder to obey the ideal gas equation?
If the molecules were not allowed to collide among themselves, would you expect more evaporation or less evaporation?
The pressure of an ideal gas is written as \[P = \frac{2E}{3V}\] . Here E refers to
The process on an ideal gas, shown in figure, is

At what temperature the mean speed of the molecules of hydrogen gas equals the escape speed from the earth?
Use R = 8.314 JK-1 mol-1
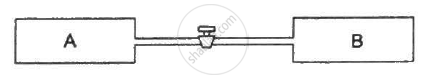
Figure shows two vessels A and B with rigid walls containing ideal gases. The pressure, temperature and the volume are pA, TA, V in the vessel A and pB, TB, V in the vessel B. The vessels are now connected through a small tube. Show that the pressure p and the temperature T satisfy `Ρ/T = 1/2 ({P_A}/{T_A}+{P_B}/{T_B))` when equilibrium is achieved.
An ideal gas is trapped between a mercury column and the closed-end of a narrow vertical tube of uniform base containing the column. The upper end of the tube is open to the atmosphere. The atmospheric pressure equals 76 cm of mercury. The lengths of the mercury column and the trapped air column are 20 cm and 43 cm respectively. What will be the length of the air column when the tube is tilted slowly in a vertical plane through an angle of 60°? Assume the temperature to remain constant.
An ideal gas is kept in a long cylindrical vessel fitted with a frictionless piston of cross-sectional area 10 cm2 and weight 1 kg in figure. The vessel itself is kept in a big chamber containing air at atmospheric pressure 100 kPa. The length of the gas column is 20 cm. If the chamber is now completely evacuated by an exhaust pump, what will be the length of the gas column? Assume the temperature to remain constant throughout the process.

Answer in brief:
Show that rms velocity of an oxygen molecule is `sqrt2` times that of a sulfur dioxide molecule at S.T.P.
Answer in brief:
Compare the rms speed of hydrogen molecules at 127ºC with rms speed of oxygen molecules at 27ºC given that molecular masses of hydrogen and oxygen are 2 and 32 respectively.
Calculate the average molecular kinetic energy
- per kmol
- per kg
- per molecule
of oxygen at 127°C, given that the molecular weight of oxygen is 32, R is 8.31 J mol−1K−1 and Avogadro’s number NA is 6.02 × 1023 molecules mol−1.
Earth’s mean temperature can be assumed to be 280 K. How will the curve of blackbody radiation look like for this temperature? Find out λmax. In which part of the electromagnetic spectrum, does this value lie? (Take Wien's constant b = 2.897 × 10−3 m K)
Calculate the energy radiated in one minute by a blackbody of surface area 200 cm2 at 127 °C (σ = 5.7 x 10-8 J m-2 s-1 K-4)
Under which condition laws of Boyle, Charles, and Gay-Lussac are valid?
Average kinetic energy of H2 molecule at 300K is 'E'. At the same temperature, average kinetic energy of O2 molecule will be ______.
Assuming the expression for the pressure exerted by the gas on the wall of the container, it can be shown that pressure is ______.
An ideal gas in a container of volume 500 cc is at a pressure of 2 × 105 N/m2. The average kinetic energy of each molecule is 6 × 10−21 J. The number of gas molecules in the container is ______.
The molecules of a given mass of a gas have root mean square speeds of 100 ms−1 at 27°C and 1.00 atmospheric pressure. What will be the root mean square speeds of the molecules of the gas at 127°C and 2.0 atmospheric pressure?
An insulated container containing monoatomic gas of molar mass m is moving with a velocity vo. If the container is suddenly stopped, find the change in temperature.
The Q-value of a nuclear reaction and kinetic energy of the projectile particle, KP are related as ______.
A proton, a deuteron and an α-particle with same kinetic energy enter into a uniform magnetic field at right angle to magnetic field. The ratio of the radii of their respective circular paths is ______.
For a particle moving in vertical circle, the total energy at different positions along the path ______.
When a particle oscillates simple harmonically, its kinetic energy varies periodically. If frequency of the particle is n, then the frequency of the kinetic energy is ______.
Assuming the expression for the pressure P exerted by an ideal gas, prove that the kinetic energy per unit volume of the gas is `3/2` P.
Show that the average energy per molecule is proportional to the absolute temperature T of the gas.
