Advertisements
Advertisements
प्रश्न
Figure shows a cylindrical tube of cross-sectional area A fitted with two frictionless pistons. The pistons are connected to each other by a metallic wire. Initially, the temperature of the gas is T0 and its pressure is p0 which equals the atmospheric pressure. (a) What is the tension in the wire? (b) What will be the tension if the temperature is increased to 2T0 ?

उत्तर
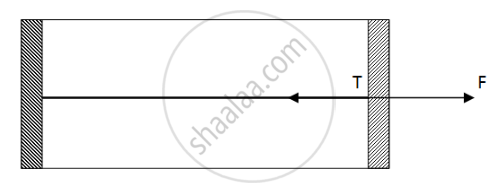
(a)
Since pressure from outside and inside the cylinder is the same, there is no net pressure acting on the pistons. So, tension will be zero.
(b)
`T_1 = T_0`
`T_2 = 2T_0`
`P_1 = P_0 = 10^5 "Pa"`
CSA = A
Let the Pistons be L distance apart .
V = AL
Applying five variable gas equation , we get
`(P_1V)/T_1 = (P_2V)/T_2`
⇒ `10^5/T_0 = P_2/(2T_0)`
⇒ `P_2 = 2×10^5 = 2P_0`
Net Force acting outside = `2P_0 - P_0 = P_0`
Force acting on a piston `F = P_0A`
By the free body diagram , we get
`F - T = 0`
`T = P_0A`
APPEARS IN
संबंधित प्रश्न
A gas is kept in an enclosure. The pressure of the gas is reduced by pumping out some gas. Will the temperature of the gas decrease by Charles's low?
The pressure of an ideal gas is written as \[P = \frac{2E}{3V}\] . Here E refers to
The temperature and pressure at Simla are 15.0°C and 72.0 cm of mercury and at Kalka these are 35.0°C and 76.0 cm of mercury. Find the ratio of air density at Kalka to the air density at Simla.
Use R=8.314J K-1 mol-1
The average translational kinetic energy of air molecules is 0.040 eV (1 eV = 1.6 × 10−19J). Calculate the temperature of the air. Boltzmann constant k = 1.38 × 10−23 J K−1.
If a = 0.72 and r = 0.24, then the value of tr is ______.
At what temperature will oxygen molecules have same rms speed as helium molecules at S.T.P.? (Molecular masses of oxygen and helium are 32 and 4 respectively).
When a gas is heated, its temperature increases. Explain this phenomenon on the basis of the kinetic theory of gases.
Find the temperature of a blackbody if its spectrum has a peak at (a) λmax = 700 nm (visible), (b) λmax = 3 cm (microwave region) (c) λmax = 3 m (short radio waves). (Take Wien’s constant b = 2.897 × 10-3 m.K).
Under which condition laws of Boyle, Charles, and Gay-Lussac are valid?
Above what temperature, all bodies radiate electromagnetic radiation?
If the density of nitrogen is 1.25 kg/m3 at a pressure of 105 Pa, find the root mean square velocity of nitrogen molecules.
What is the microscopic origin of temperature?
Explain in detail the kinetic interpretation of temperature.
Two molecules of a gas have speeds of 9 × 10 6 ms−1 and 1 × 106 ms−1, respectively. What is the root mean square speed of these molecules?
An insulated container containing monoatomic gas of molar mass m is moving with a velocity vo. If the container is suddenly stopped, find the change in temperature.
Explain why there is no atmosphere on moon.
Assuming the expression for the pressure P exerted by an ideal gas, prove that the kinetic energy per unit volume of the gas is `3/2` P.
If a = 0. 72 and r = 0.24, then the value of t is ______.
2000 calories of radiant heat is incident on a body. If the body absorbs 550 calories of heat, find the coefficient of emmission of the body.
