Advertisements
Advertisements
प्रश्न
If a = 0.72 and r = 0.24, then the value of tr is ______.
विकल्प
0.02
0.04
0.4
0.2
उत्तर
If a = 0.72 and r = 0.24, then the value of tr is 0.04.
Explanation:
Express the relation of absorptivity. The fraction of light/ray/radiation absorbed by a surface is called absorptivity.
`a = (G_(abs))/G`
`G_(abs) =Ga`
Here, `G_(abs)` is absorbed radiation and G is incident radiation.
Express the relation of reflectivity.
The fraction of light reflected by a surface is known as reflectivity.
`r = (G_(ref))/G`
`G_(ref) = Gr`
Here, `G_(ref)` is reflected radiation.
Express the relation of transmissivity. The fraction of light/ray/radiation transmitted by a surface is known as transmissivity.
`t_r = G_t/G`
`G_t = Gt_r`
Here, `G_t` is transmitted radiation.
Express the relation of 1st law of thermodynamics. Sum of all radiation (absorbed, reflected and transmitted) should be equals to the total radiation.
`G_(abs) + G_(ref) + G_t = G` ......(i)
Substitute `Ga` for `G_(abs), G_r` for `G_(mf)` and Gt for Gt in equation (i).
`(Ga)/G + (Gr)/G + (Gt_r)/G` = 1
`a + r + t_r` = 1
Substitute 0.72 for a and 0.24 for r to find transmitted radiation.
0.72 + 0.24 + tr = 1
tr = 1 – 0.96
tr = 0.04
APPEARS IN
संबंधित प्रश्न
Is it possible to boil water at room temperature, say 30°C? If we touch a flask containing water boiling at this temperature, will it be hot?
Which of the following quantities is the same for all ideal gases at the same temperature?
(a) The kinetic energy of 1 mole
(b) The kinetic energy of 1 g
(c) The number of molecules in 1 mole
(d) The number of molecules in 1 g
Find the number of molecules of an ideal gas in a volume of 1.000 cm3 at STP.
A gas cylinder has walls that can bear a maximum pressure of 1.0 × 106 Pa. It contains a gas at 8.0 × 105 Pa and 300 K. The cylinder is steadily heated. Neglecting any change in the volume, calculate the temperature at which the cylinder will break.
During an experiment, an ideal gas is found to obey an additional law pV2 = constant. The gas is initially at a temperature T and volume V. Find the temperature when it expands to a volume 2V.
Use R = 8.3 J K-1 mol-1
One mole of an ideal gas undergoes a process `P = (P_0)/(1+(V/V_0)^2` where `p_0` and `V_0` are constants . Find the temperature of the gas when `V=V_0` .
The weather report reads, "Temperature 20°C : Relative humidity 100%". What is the dew point?
The condition of air in a closed room is described as follows. Temperature = 25°C, relative humidity = 60%, pressure = 104 kPa. If all the water vapour is removed from the room without changing the temperature, what will be the new pressure? The saturation vapour pressure at 25°C − 3.2 kPa.
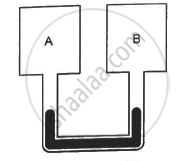
Figure shows two rigid vessels A and B, each of volume 200 cm3, containing an ideal gas (Cv = 12.5 J K−1 mol−1). The vessels are connected to a manometer tube containing mercury. The pressure in both the vessels is 75 cm of mercury and the temperature is 300 K. (a) Find the number of moles of the gas in each vessel. (b) 5.0 J of heat is supplied to the gas in vessel A and 10 J to the gas in vessel B. Assuming there's no appreciable transfer of heat from A to B, calculate the difference in the heights of mercury in the two sides of the manometer. Gas constant, R = 8.3 J K−1 mol−1.
Using figure, find the boiling point of methyl alcohol at 1 atm (760 mm of mercury) and at 0.5 atm.

Answer in brief:
What will happen to the mean square speed of the molecules of a gas if the temperature of the gas increases?
Answer in brief:
Compare the rms speed of hydrogen molecules at 127ºC with rms speed of oxygen molecules at 27ºC given that molecular masses of hydrogen and oxygen are 2 and 32 respectively.
When a gas is heated, its temperature increases. Explain this phenomenon on the basis of the kinetic theory of gases.
Calculate the average molecular kinetic energy
- per kmol
- per kg
- per molecule
of oxygen at 127°C, given that the molecular weight of oxygen is 32, R is 8.31 J mol−1K−1 and Avogadro’s number NA is 6.02 × 1023 molecules mol−1.
The emissive power of a sphere of area 0.02 m2 is 0.5 kcal s-1m-2. What is the amount of heat radiated by the spherical surface in 20 seconds?
Earth’s mean temperature can be assumed to be 280 K. How will the curve of blackbody radiation look like for this temperature? Find out λmax. In which part of the electromagnetic spectrum, does this value lie? (Take Wien's constant b = 2.897 × 10−3 m K)
Under which condition laws of Boyle, Charles, and Gay-Lussac are valid?
Why the temperature of all bodies remains constant at room temperature?
A metal cube of length 4 cm radiates heat at the rate of 10 J/s. Find its emissive power at a given temperature.
Average kinetic energy of H2 molecule at 300K is 'E'. At the same temperature, average kinetic energy of O2 molecule will be ______.
An inflated rubber balloon contains one mole of an ideal gas, has a pressure p, volume V and temperature T. If the temperature rises to 1.1 T, and the volume is increased to 1.05 V, the final pressure will be ______.
A gas mixture consists of molecules of types A, B and C with masses mA > mB > mC. Rank the three types of molecules in decreasing order of average K.E.
A gas mixture consists of molecules of types A, B and C with masses mA > mB > mC. Rank the three types of molecules in decreasing order of rms speeds.
The Q-value of a nuclear reaction and kinetic energy of the projectile particle, KP are related as ______.
A proton, a deuteron and an α-particle with same kinetic energy enter into a uniform magnetic field at right angle to magnetic field. The ratio of the radii of their respective circular paths is ______.
Assuming the expression for the pressure P exerted by an ideal gas, prove that the kinetic energy per unit volume of the gas is `3/2` P.
According to the kinetic theory of gases, at a given temperature, molecules of all gases have the same ______.
If a = 0. 72 and t = 0.04, then the value of r is ______.
