Advertisements
Advertisements
प्रश्न
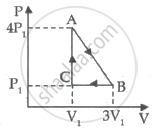
50 cal of heat should be supplied to take a system from the state A to the state B through the path ACB as shown in figure. Find the quantity of heat to be suppled to take it from A to B via ADB.

उत्तर
Given:-
In path ACB,
∆Q = 50 cal = (50 × 4.2) J
∆Q = 210 J
∆W = WAC + WCB
Since initial and final volumes are the same along the line BC, change in volume of the system along BC is zero.
Hence, work done along this line will be zero.
For line AC:-
P = 50 kPa
Volume changes from 200 cc to 400 cc.
`rArrDelta V =400-200c c=200 c c`
∆W = WAC + WCB
= 50 × 10−3 × 200 × 10−6 + 0
= 10 J
Using the first law of thermodynamics, we get
∆Q = ∆U + ∆W
⇒ ∆U = ∆Q − ∆W = (210 − 10) J
∆U = 200 J
In path ADB, ∆Q = ?
∆U = 200 J ..............(Internal energy depends only on the initial and final points and not on the path followed.)
⇒ ∆W = WAD + WDB = ∆W
Work done for line AD will also be zero.
For line DB:-
P = 155 kPa
`rArrDelta V =400-200c c=200 c c`
W = 0 + 155 × 103 × 200 × 10−6
W = 31 J
∆Q = ∆U + ∆W
∆Q= (200 + 31) J = 231 J
∆Q = 55 cal
APPEARS IN
संबंधित प्रश्न
When we heat an object, it expands. Is work done by the object in this process? Is heat given to the object equal to the increase in its internal energy?
The pressure of a gas changes linearly with volume from 10 kPa, 200 cc to 50 kPa, 50 cc. (a) Calculate the work done by the gas. (b) If no heat is supplied or extracted from the gas, what is the change in the internal energy of the gas?
Answer the following in one or two sentences.
State the first law of thermodynamics.
A mixture of hydrogen and oxygen is enclosed in a rigid insulating cylinder. It is ignited by a spark. The temperature and pressure both increase considerably. Assume that the energy supplied by the spark is negligible, what conclusions may be drawn by application of the first law of thermodynamics?
For an Isochoric process
When heat energy of 2000 joules is supplied to a gas at constant pressure 2.1 x 105 N/m2, there is an increase in its volume equal to 2.5 x 10-3 m3. The increase in internal energy of the gas in joules is ____________.
"The mass and energy both are conserved in an isolated system", is the statement of ______.
The process, in which no heat enters or leaves the system, is termed as ____________.
120 J of heat is added to a gaseous system, whose internal energy change is 60 J, then the amount of external work done is ____________.
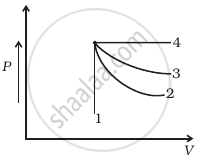
An ideal gas undergoes four different processes from the same initial state (figure). Four processes are adiabatic, isothermal, isobaric and isochoric. Out of 1, 2, 3 and 4 which one is adiabatic.
Air pressure in a car tyre increases during driving. Explain.
The first law of thermodynamics is concerned with the conservation of ______.
An ideal gas is taken through series of changes ABCA. The amount of work involved in the cycle is ______.
Which among the following equations represents the first law of thermodynamics under isobaric conditions?
If one mole of monoatomic gas `(gamma=5/3)` is mixed with one mole of diatomic gas `(gamma=7/5)`, the value of γ for the mixture is ______.
ΔU = 0 is true for ______.
The amount of work done in increasing the voltage across the plates of capacitor from 5 V to 10 V is W. The work done in increasing it from 10 V to 15 V will be ______.
In an adiabatic expansion of 2 moles of a gas, the initial pressure was 1.013 × 105 Pa, the initial volume was 22.4 L, the final pressure was 3.191 × 104 Pa and the final volume was 44.8 L. Find the work done by the gas on its surroundings. Taken `γ = 5/3`.
What is Isobaric process?
Define the isothermal process.
