Advertisements
Advertisements
प्रश्न
A 100 kg lock is started with a speed of 2.0 m s−1 on a long, rough belt kept fixed in a horizontal position. The coefficient of kinetic friction between the block and the belt is 0.20. (a) Calculate the change in the internal energy of the block-belt system as the block comes to a stop on the belt. (b) Consider the situation from a frame of reference moving at 2.0 m s−1 along the initial velocity of the block. As seen from this frame, the block is gently put on a moving belt and in due time the block starts moving with the belt at 2.0 m s−1. calculate the increase in the kinetic energy of the block as it stops slipping past the belt. (c) Find the work done in this frame by the external force holding the belt.
उत्तर
Here,
m = 100 kg
u = 2.0 m/s
v = 0
μk = 0.2
a) Internal energy of the belt-block system will decrease when the block will lose its KE in heat due to friction. Thus,
KE lost = `1/2m u^2-1/2mnu^2`
`1/2m(u^2-nu^2)`
`1/2xx100xx(2^2-0^2)`
= 200 J
b) Velocity of the frame is given by
uf = 2.0 m/s
u’ = u - uf = 2 – 2= 0
v’ = 0 – 2 = -2 m/s
KE lost = `1/2m u'^2-1/2mnu'^2`
`=1/2m(0^2-nu'^2)`
`=1/2xx100xx(0^2-2^2)`
= 200 J
c) Force of friction is given by
`f=mu_k^""R`
`rArrf=0.2xxmg=0.2xx100xx10=200N
`"Retardation"=f/m=200/100=2ms^-2`
Distance moved by the block will be as seen from the frame = s
`nu'^2-u'^2=2as`
`rArr2^2-0^2=2xx2s`
`rArrs=1"m"`
Work done by the force responsible for accelaration as seen from the frame = fs
= 200 x 1 = 200J
Work done by the belt to give it a final velocity of 2 m/s
`=1/2mnu'^2`
`=1/2xx100xx(2)^2`
= 200J
Total work done by external force as seen from the frame 200 + 200 = 400J
APPEARS IN
संबंधित प्रश्न
A cylinder containing a gas is lifted from the first floor to the second floor. What is the amount of work done on the gas? What is the amount of work done by the gas? Is the internal energy of the gas increased? Is the temperature of the gas increased?
When we rub our hands they become warm. Have we supplied heat to the hands?
Can work be done by a system without changing its volume?
Consider the following two statements.
(A) If heat is added to a system, its temperature must increase.
(B) If positive work is done by a system in a thermodynamic process, its volume must increase.
The pressure p and volume V of an ideal gas both increase in a process.
(a) Such a process is not possible.
(b) The work done by the system is positive.
(c) The temperature of the system must increase.
(d) Heat supplied to the gas is equal to the change in internal energy.
In a process on a system, the initial pressure and volume are equal to the final pressure and volume.
(a) The initial temperature must be equal to the final temperature.
(b) The initial internal energy must be equal to the final internal energy.
(c) The net heat given to the system in the process must be zero.
(d) The net work done by the system in the process must be zero.
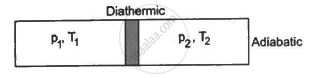
Figure shows a cylindrical tube of volume V with adiabatic walls containing an ideal gas. The internal energy of this ideal gas is given by 1.5 nRT. The tube is divided into two equal parts by a fixed diathermic wall. Initially, the pressure and the temperature are p1, T1 on the left and p2, T2 on the right. The system is left for sufficient time so that the temperature becomes equal on the two sides. (a) How much work has been done by the gas on the left part? (b) Find the final pressures on the two sides. (c) Find the final equilibrium temperature. (d) How much heat has flown from the gas on the right to the gas on the left?
A system releases 130 kJ of heat while 109 kJ of work is done on the system. Calculate the change in internal energy.
Define heat.
When does a system lose energy to its surroundings and its internal energy decreases?
One gram of water (1 cm3) becomes 1671 cm3 of steam at a pressure of 1 atm. The latent heat of vaporization at this pressure is 2256 J/g. Calculate the external work and the increase in internal energy.
A thermodynamic system goes from states
(i) P, V to 2P, V (ii) P, V to P, 2V
The work done in the two cases is ____________.
An ideal gas is compressed at a constant temperature. Its internal energy will ____________.
In a thermodynamic system, working substance is ideal gas. Its internal energy is in the form of ______.
Two cylinders A and B of equal capacity are connected to each other via a stopcock. A contains a gas at standard temperature and pressure. B is completely evacuated. The entire system is thermally insulated. The stopcock is suddenly opened. Answer the following:
What is the change in internal energy of the gas?
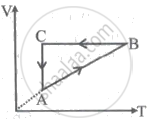
A cyclic process ABCA is shown in the V-T diagram. A process on the P-V diagram is ______.
The internal energy of one mole of argon is ______.
A steam engine delivers 4.8 x 108 Jof work per minute and services 1.2 x 109 J of heat per minute from its boiler. What is the percentage efficiency of the engine?
What is heat?
