Advertisements
Advertisements
Question
Consider the following cyclic process consist of isotherm, isochoric and isobar which is given in the figure.
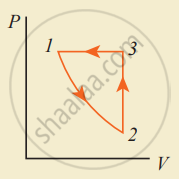
Draw the same cyclic process qualitatively in the V-T diagram where T is taken along the x-direction and V is taken along the y-direction. Analyze the nature of heat exchange in each process.
Solution
Process 1 to 2: increase in volume. So heat must be added.
Process 2 to 3: Volume remains constant. Increase in temperature. The given heat is used to increase the internal energy.
Process 3 to 1: Pressure remains constant. Volume and Temperature are reduced. Heat flows out of the system. It is an isobaric compression where the work is done on the system.

Explanation: In the graph, during the process (1 to 2), the gas undergoes isothermal expansion. It receives a certain amount of heat from its surroundings. It uses this heat in doing the work. Hence internal energy of the gas remains unchanged.
During the process represented by (2 to 3), the gas is heated at a constant volume. Since no work is done and volume does not change, the process is an isochoric process.
Since heat is transferred to the gas from the surroundings, the internal energy of the gas is increased. During the process represented by (3 to 1), the gas is compressed isobarically. Work is done on the gas. Since temperature drops internal energy is reached. Hence the gas gives up heat to the surroundings.
APPEARS IN
RELATED QUESTIONS
A thermodynamic system is taken from an original state to an intermediate state by the linear process shown in Figure

Its volume is then reduced to the original value from E to F by an isobaric process. Calculate the total work done by the gas from D to E to F
For work done to be reversible, the process should be ______
Draw a p-V diagram of the irreversible process.
Draw a p-V diagram showing negative work with varying pressure.
Give the equation of state for an isothermal process.
Give an equation state for an isochoric process.
Derive the work done in an adiabatic process.
An ideal gas is expanded isothermally from volume V1 to volume V2 and then compressed adiabatically to original volume V1. If the initial pressure is P1, the final pressure is P3 and net work done is W, then ____________.
An ideal gas is compressed to half its initial volume by means of several processes. Which of the process results in the maximum work done on the gas?
When an inflated ballon is suddenly burst, why is the emerging air slightly cooled?
